Learn smarter with fresh insights from our tutors.
Practical study guides, course supplements, and learning resources curated to help you move faster through every subject.
Diabetes As a Side Effect of Cancer Treatment

Diabetes As a Side Effect of Cancer Treatment
As if battling cancer isn't enough, many patients also have to deal with the unexpected side effect of diabetes during and after treatment. This can be a daunting experience for anyone, but knowing what to expect and how to manage it can make a huge difference in your overall health and well-being.
In this blog post, we'll cover important things you should know about diabetes as a potential side effect of cancer treatment, so you can face this challenge head on with confidence and knowledge. Whether you're a patient, caregiver or simply interested in learning more about this topic – keep reading!
Cancer Treatments That Can Cause Diabetes
Cancer treatments that can cause diabetes include chemotherapy, radiation therapy, and certain targeted cancer drugs. These treatments can damage the pancreas, leading to diabetes. People who have cancer are at an increased risk for developing diabetes, especially if they have a family history of the condition.
If you have cancer and are treated with any of these therapies, your doctor will closely monitor your blood sugar levels. It’s important to keep your blood sugar under control because high levels can lead to serious complications such as heart disease, stroke, and kidney disease.
There are some things you can do to help lower your risk of developing diabetes after cancer treatment. Maintaining a healthy weight, eating a healthy diet, and getting regular exercise can all help reduce your risk. If you smoke, quitting smoking is also important in lowering your risk of developing diabetes.
Symptoms of Diabetes
Patients with cancer should be cognizant of some key indications of diabetes. These include excessive thirst, increased frequency in urination, exhaustion and unexpected weight loss. If you recognize any of these signs, it is essential to talk to your doctor immediately for testing and possible treatment.
Risks Associated With Diabetes
There are a number of risks associated with diabetes, some of which can be serious or even life-threatening. These include:
- Diabetic ketoacidosis – a condition that can occur if insulin levels drop too low, leading to the build-up of ketones in the blood which can lead to coma or death
- Diabetic neuropathy – damage to the nerves caused by high blood sugar levels, which can lead to pain, numbness or paralysis in the extremities
- Cardiovascular disease – diabetes is a major risk factor for heart disease and stroke
- Kidney disease – diabetes is the leading cause of kidney failure
- Eye problems – including diabetic retinopathy, which can lead to blindness
People with diabetes need to be aware of these risks and take steps to minimise them, such as keeping their blood sugar levels under control and attending regular check-ups with their doctor.
How to Manage Diabetes
There are a few different ways to manage diabetes, depending on its severity. These include lifestyle changes (such as diet and exercise), medication, and insulin therapy. Your doctor will work with you to create a management plan that is right for you.
In conclusion, diabetes is a challenging complication that sometimes arises as a result of cancer treatments. With the right knowledge and help on hand, those with diabetes brought on by such treatments can successfully address their symptoms and keep their health in check. We trust that this article has enabled you to gain greater insight into why the treatment of cancer can lead to diabetes and strategies one may employ for managing it. Remember: assistance can be accessed, so don't hesitate to seek it out!
Atomic Theory and the Structure of the Atom
Atomic Theory and the Structure of the Atom
Learning Outcomes
By the end of this article, you should be able to
- Define a theory
- Describe the structure of the atom.
- Calculate relative atomic mass of an atom
- Define elements and compounds
- Calculate relative molecular and formula masses of compounds
- Write electronic structures of elements
- Explain pure substances and mixtures
- Describe the periodic table of elements and their properties
What is a Theory
Scientific problems are always checked by experimental work. If a large number of experiments all give similar results, the latter are then summarized into a single statement known as a law.
Thus, a law is a summary of the results of a large number of separate experiments all giving the same conclusion. Further experiments are carried out to account for the law. This is done by proposing a hypothesis.
If the results of the experiments agree with the hypothesis, those results are stated into what is called a theory.
Atomic Theory
The most important chemical theory was given by John Dalton (1766-1844). Initially, Dalton stated the hypothesis that all substances consist of small particles of matter of several differentkinds corresponding to the different elements.
He called these particles atoms (from Greek: 'atomos' which means indivisible).
Dalton also said that elements consist of atoms and that atoms of one element are identical and compounds result from the combination of atoms of two or more elements each in a definite number.
He assigned a weight of 1 (one) to a hydrogen atom and built a table of relative weights of elements and compounds.
This hypothesis was verified by further experimental work and so it became a theory. Thus Dalton's atomic theory can be summarized into the following statements:
a) All matter is made up of atoms that cannot be created or destroyed; that is, atoms are indivisible by ordinary chemical means
b) Atoms of a particular element are identical in size, shape, mass and all other properties and differ from other atoms in these properties.
Experiments that were carried out later showed that atoms of one element may have different masses depending on the number of neutrons present.
Atoms of one element having different masses are called isotopes. You will learn more about isotopes. For now you should realize that statement (b) is not very correct.
Structure of the Atom
In Science, people are always curious. Having accepted that an atom exists, the Scientists then wanted to know how this atom looks like. As such, they carried out several studies.
From these studies, it was found that the atom has even smaller particles which are called subatomic particles.
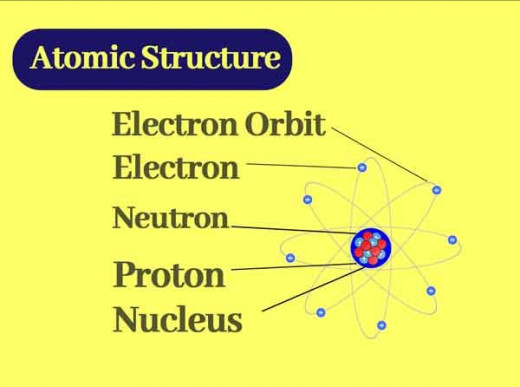
For example it was found that an atom consists of a positively charged nucleus surrounded by light (not heavy) negatively charged particles called which were named electrons.
In 1909, Ernest Rutherford studied the deflection patterns of alpha particles (He2+ ions) by thin metal foils and an analysis of the results led him to the conclusion that an atom has a tiny central nucleus that carries practically all the mass of the atom and that the lighter particles (the electrons) are around this nucleus. The number of positive charges on the nucleus is called the atomic number (abbrev. Z).
However, since the atom was found to be neutral, it was clear that the number of positively charged particles must equal the number of negatively charged particles (electrons).
Therefore, the number of electrons surrounding the nucleus in a neutral atom, must also be equal to the atomic number. The positively charged particles are called protons. The hydrogen atom for example, has one proton in its nucleus and carries one electron.
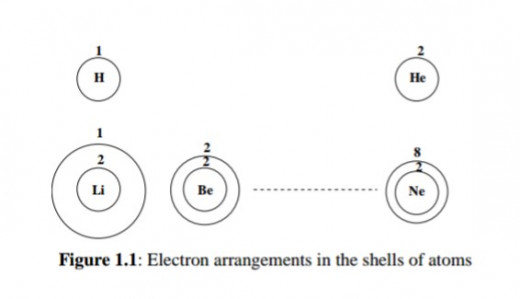
Furthermore, it was discovered that the electrons are arranged around the nucleus in ''shells''. Imagine this on a magnified scale.
You put the first group of electrons on a small ''circle'' with the nucleus inside it. Then put the second group of electrons on a bigger circle with the smaller one inside it and so on.
Figure 1.1 illustrates this pattern.
Electron Arrangements in the Shells of Atoms
Thus the successively added electrons first fill the innermost shell, then the next shell to it and so on. The innermost shell is called the K shell, the other shells in order of increasing size are called L, M, N, and so on.
We shall see later that the K shell is the lowest in energy, the L shell is higher in energy than the K shell, the M shell is higher in energy than the L shell and so on. We will advance the structure of the atom later.
Relative Atomic Mass (RAM)
We have stated that an atom has some components. The electrons are negatively charged and each has a charge of -1.60206 × 10-19 coulombs and a mass of 9.109 × 10-28g. The electron charge is abbreviated 'e' (-1.60206 ×10-19 = e) so that one electron has a charge of -e or just -1.
It should be noted here that the minus (-) sign does not mean that the electron is short of electric charge but that its electrical charge is of the type called negative. The protons are positively charged and each has a charge of +1.60206 × 10-19 coulombs and a mass of 1.672 × 10-24g.
As for the electron, the charge on a proton is simplified to +1. All the atoms except hydrogen also have neutrons which are neutral (have a charge of zero) but have a mass of 1.675 × 10-24g. The other components of an atom include neutrinos, positrons, μ-mesons and pi-mesons among others but the detailed nature of these is beyond the scope of this article.
The absolute masses of atoms cannot be determined as the atoms are very light. Only the relative masses can be obtained. To get these relative masses, we need to assign a weight to one atom (the standard) and relate the masses of the others to it. What we get then is the Relative Atomic Mass (RAM) of the atom.
Initially, hydrogen which had been assigned a mass of one, was chosen as the standard. However, for some practical reasons a carbon atom with six protons and six neutrons (carbon-12) was later chosen as the new standard.
The carbon-12 has a mass of exactly 12. The atomic masses of all the atoms were obtained with reference to a carbon-12 as the standard.
However, with technological revolution in modern times, mass spectrometers are used to determine the relative masses of atoms directly. Note that the relative atomic masses are rations hence have no units.
Since there are more than one atom for a particular element with differing relative masses we need to calculate a weighted mean of their masses taking into account their relative abundances.
This weighted mean for the mass of a particular element is called the atomic weight of that element. Therefore, we define the atomic weight of an element as the weighted mean of the relative masses of the isotopes of that particular element.
For example, chlorine has two isotopes with the relative masses and abundances of 35 (75.5%) and 37 (24.5%). Thus the atomic weight (the relative atomic mass) of chlorine is:
[ (35 × 75.5)/100] + [(37×24.5)/100]=35.4 9
Note in the example above that there is no chlorine in nature with a mass of 35.49. The atomic unit is called the Atomic mass unit (amu, given the SI unit, μ). One atomic mass unit is equivalent to1.66×10-24g (i.e., 1 amu = 1.66×10-24g ). Using this basis of measurement, the mass of an electron is 9.109×10-28g ÷1.66×10-24 g/amu = 0.000548 amu and that of a proton is 1.672×10-24g ÷ 1.66×10-24g/amu = 1.00725 amu and that of a neutron is 1.675×10-24 g ÷ 1.66×10-24g/amu = 1.00861 amu.
We usually round off atomic mass units to whole numbers called Mass Numbers (abbrev. A). This means that the mass of an electron is 0 while that a proton and a neutron each has a mass of 1.
The mass number (A) of an atom is the sum total of the number of protons and neutrons (since electrons are so light their contribution to the total mass is ignored). If we abbreviate the mass of neutrons N, then we have:
A =Z + N
where A, is the mass number, Z is the number (= mass)of protons
and N is the number (= mass) of neutrons.
It is possible to calculate the weight of an atom in grams or kilograms. Suppose, for example that 6×10-23 hydrogen atoms weigh 1g, what is the weight of one hydrogen atom?
Simple proportion shows that one hydrogen atom weighs 1g ÷ 6×1023 = 1.66×10-24g.
Note that the value obtained equals the weight of a single proton as given earlier. Since one hydrogen atom also weighs 1 amu, this confirms the fact that 1 amu= 1.66×10-24g as given above.
However, we don’t usually express atomic weights in grams or kilograms as the values become cumbersome in metric units.
Since 6 ×10-23 hydrogen atoms weigh 1g and one hydrogen atom weighs 1 amu, it follows that 1g = 6 ×1023amu. The figure 6×1023 (or more correctly 6.0226×1023) can be used to convert grams to atomic mass units. It is called the Avogadro's Number.
Isotopes
Atoms of a particular element all have the same atomic number and therefore, the same number of protons. Most of the elements do not have all their atoms with the same number of neutrons.
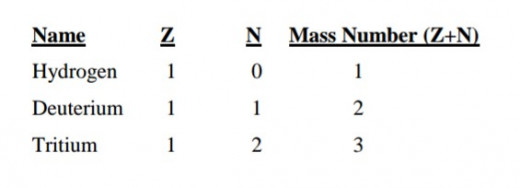
Such atoms belonging to the same element but having different numbers of neutrons in their nuclei are called isotopes.
It is clear therefore that isotopes have different masses. This is indeed the case as shown by the example below for the isotopes of hydrogen.
Elements and Compounds
Table 1.1 shows the names of some elements and their symbols.
Table 1.1: Some elements and their symbols

Some Elements and Their Symbols
Note that the first letter in the name of some few elements is taken as a symbol of that particular element. For example, hydrogen is H, carbon is C, nitrogen is N and so on.
However, because there are many more elements than the number of letters in the alphabet, some elements have two letters in their symbols. For these, the first letter is always in capitalized and the second one is written in small letter.
For example, chlorine is Cl, magnesium is Mg, neon is Ne and so on. In the foregoing examples, the first letter is derived from the English name. In other cases, the symbols of the elements are derived from Latin or German names.
For example, Antimony is Sb (from Latin, Stibnium), Copper is Cu (from Latin, Cuprum), Iron is Fe (from Latin, Ferrum), Sodium is Na (from German, Natrium), and potassium is K (from German, Kalium), sodium is Na (from German, Natrium) and potassium is K (from German, Kalium).
A compound is formed when atoms of two or more different elements combine. Examples of some compounds include water (H2O), methane (CH4), glucose (C6H12O6) and so on.
The kind and number of atoms in a compound are described by a formula as shown in brackets after the names of those compounds.
The usage of a formula can also be extended to an element if it naturally exists as a molecule. An example is dichloride (Cl2).
A formula indicates the relative numbers of atoms in a substance. There are three types of formulae; empirical formula is the simplest and gives a bare minimum information about a compound.
Diamond for example, consists of an array of carbon atoms each covalently bonded to four other carbon atoms but the formula of diamond, C, does not indicate this.
The other empirical formulae include, among others, NaCl (sodium chloride) and SiC (silicon carbide). In the general empirical formula of a compound, AxBy (where A and B are the atoms combined), the subscripts x and y are usually unity.
The second is a molecular formula, which has more information about a compound than an empirical formula. In the general formula AxDy, (where A and D are the atoms combined), the subscripts x and y in a molecular formula, are the actual numbers of atoms in the compound. For example, water has two hydrogen atoms and one oxygen atom; its molecular formula is therefore H2O.
The third type is a structural formula which shows how the atoms are joined to each other. In this case, the dashes are used to represent bonds and the atomic symbols represent atoms. For example, the structural formula of water is H-O-H.
Relative Molecular and Formula Mass
In the Section above, it was mentioned that compounds that exist as discrete entities, are described by a molecular formula. Such compounds are called molecules. Since the masses of atoms are relative, the masses of molecules are also relative.
Each molecule has its own relative mass which is the sum of the relative atomic masses of the atoms in that molecule. We therefore define the Relative Molecular Mass (RMM) as the sum of the relative masses of the atoms in a molecular formula.
For example:
(a) H2O has two hydrogen (H) atoms and one oxygen (O) atom
Relative molecular mass of water = (2× relative atomic mass of H) + (relative atomic mass of O)
= (2 × 1) + 16
= 18
(b) N2 has two Nitrogen atoms
Relative molecular mass of nitrogen molecule = (2× relative atomic mass of N) = 2 × 14
= 28
(c )C6H12O6 (glucose) has six carbon (C) atoms, 12 hydrogen (H) atoms and 6 oxygen (O) atoms
Relative molecular mass of glucose = (6 × relative atomic mass of C)
+(12 × relative atomic mass of H + (6 × relative atomic mass of O)
= 72 + 12 + 96
= 180
Compounds that do not exist as discrete entities however, do not form molecules and are represented by empirical formula.
For these we do not assign a Relative Molecular Mass; instead a Relative Formula Mass (RFM) is used. The relative formula mass is defined as the sum of the relative masses of the atoms in an empirical formula. For example:
NaCl has one sodium (Na) atom and one chlorine (Cl) atom
Relative formula mass of NaCl = (1× relative atomic mass of Na + (1× relative atomic mass of Cl)
= (1×23) + (1×35.5)
= 58.5
Pure Substances and Mixtures
We define substance that contains only one kind of compound or element as a pure substance. Take a spoon full of sugar, for example, this spoon contains only sugar molecules; therefore sugar is a pure substance.
Similarly, take copper metal; this consists of only copper atoms. Copper is also a pure substance. Pure substances have got the same, definite composition. This means that the concentration of the substance in a pure compound is constant.
When two or more different compounds (or elements) are put together and cannot react with each other, then we have a mixture.
Air for example, contains oxygen, nitrogen, water, carbon dioxide among others. It is a mixture of gases. Unlike pure substances, mixtures do not have definite compositions. The composition of substances in air, for example, may vary depending on weather conditions.
There are two types of mixtures; those in which the particles of each component are visibly present are called heterogeneous mixtures. An example is a mixture of say, salt and black pepper.
In this mixture, you can see the salt particles (white) and the pepper particles (black). The second type of mixture is where the particles of one component are evenly dispersed amongst those of the other.
This is called a homogeneous mixture. It is also called solution. Sugar or salt dissolved in water; methanol mixed with water; air on its own, are some examples of solutions.
Quantum Theory
We started on the subject of atomic structure in Section 1.3. We will now go a step further with this subject in this section. It is common experience than when you hit a football, it will move to any distance depending on the amount of energy you give it. Consider Figure 1.2a as an example.
The ball which is hit from position A, can stop at any of the dark points indicated. In this case, we say that the energy of the ball is continuous; That is, the ball can have any amount of energy.
If you hit the ball with less force it will gain less energy and so will move a short distance, if you hit it with more force it will gain more energy and likewise move a longer distance.
The motion of the ball here, is a part of the physics called classical physics.Newton'sLaws of motion are part of this physics. In reality, laws of classical physics apply to large particles such as the ball, the motion of the earth round the sun, automobiles, etc.
Figure 1.2: Comparison of a football having any amount of energy (a) (classical physics) and another one having only enough energy to take it from one point to the next (b)(quantum
physics)
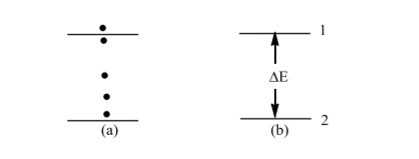
When classical physics was applied to atoms and other smaller particles, the results obtained were contrary to those expected. In 1900, Max Planck solved this mystery by stating that the energy of an object is not continuous; and that the object will only acquire enough energy to take it from one energy level to another.
Figure 1.2b illustrates this. If we give energy to an object in state 2 the object will only move to state 1 if it has acquired enough energy equal to ΔE = E1-E2(where, ΔE is change in energy, E2 is the energy of state 2 and E1 that of state 1).
Note in this case, the energy is in form of a packet or bundle and so, we speak of the energy being quantized. Thus the object can only have a certain definite energy value. Planck called this packet of energy (=ΔE) a quantum of energy.
If the energy of the object in energy level 2 is less than ΔE, the object will not move. This then started a new branch of physics called quantum physics whose laws apply to tiny particles like atoms, electrons, etc.
In 1013, NeilsBohr applied quantum theory to motion of electrons in atoms. He said that electrons move around the nucleus in circular orbits and as they do so, they emit or absorb energy that is quantized (it is now known that electrons do not actually behave this way).
The energy of an electron is governed by an integer called the Principal Quantum Number (n). The principal quantum numbers are just arbitrary numbers which are introduced in order to make predictions match experiment. They are given numbers 1,2,3,4, etc, increasing as an electron moves further away from the nucleus. In section above, we stated that electrons are in shells.
In simple terms, the principal quantum numbers are the shell numbers; The shells are numbered starting from the inter most as 1 and proceed as you move away. Thus the K shell is principal quantum number 1, L shell is principal quantum number 2 and so on.
Now the results of the experiments carried out later showed that under certain circumstances an electron behaves as a particle and under other circumstances it behaves as a wave just like a water wave. You will realise that one cannot locate the position of a wave since a wave extends in space.
We can only state that the chance of finding the wave at a particular point will be this much. In the same way, an electron being a wave, we can only state the chance of finding it at a certain place. The modern quantum theory says that electrons move around the nucleus in a definite region of space.
The region of space in which the electron spends at least 95% of its time is called an orbital. The orbitals are arranged in shells around the nucleus. For each principal quantum number (n), there are n2 orbitals.
These are shown in Table 1.2. Remember that numbering starts from the innermost shell since the K shell is shell number 1(that is principal quantum number 1).
Table 1.2: Shell symbols, orbitals and the maximum number of electrons in them for each principal quantum number
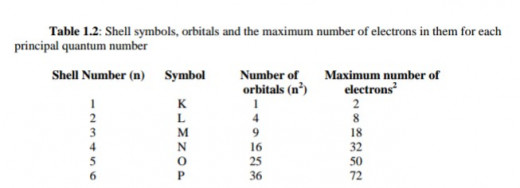
Shell Symbols, Orbitals and Maximum Number of Electrons
Apart from the K shell which has one orbital (the s orbital), both theory and experiments have shown that the group of orbitals in each of the L, M, N, etc., shells do not have the same energies especially in a many-electron atom.
In a hydrogen atom, which has only one electron, the orbitals in each group have the same energy. Such orbitals having the same energy are said to be degenerate. In a many-electron atom, the L shell has two energy levels; the lower one has one orbital which is called an s orbital, the higher one has three identical orbitals which are degenerate and these are called p orbitals.
The s orbital is spherical whereas the p orbitals are dumb-bell shaped situated at right angles to each other and in three dimensions along the x-, y- and z-axes; hence designated npx, npy and npz respectively, (where n is the principal quantum number).
The M shell has three energy levels; the lowest one consists of one orbital (also an s orbital), the second one has three orbitals which are identical and degenerate (also called p orbitals); and the third level consists of five degenerate orbitals called d orbitals. The N shell has four energy levels, s, p and d just like the M shell, but in addition, the highest one has seven degenerate orbitals called f orbitals.
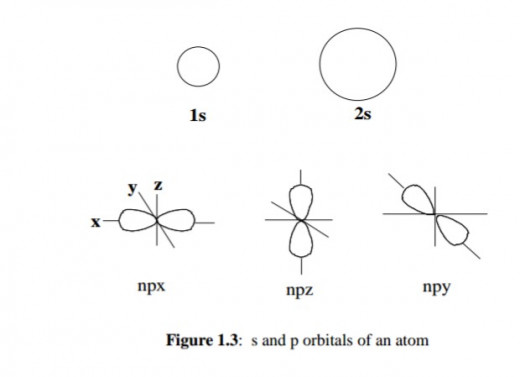
s and p Orbitals of an atom
Therefore, within a given shell, the energies increase in the order s<p<d<f3, etc. It was stated earlier that the energy of the electron is governed by the principal quantum number n; this number also governs the size of the orbitals.
As shown in Figure 1.3, an s orbital of principal quantum number one (denoted 1s) is smaller than an s orbital of principal quantum number two (denoted 2s).
As we shall see in the next section, the orbitals can be specified by the ''n'' values in which they appear; thus we would talk of a 1s orbital to mean an s orbital of principal quantum number one.
Similarly, a 2s or 3s orbital would mean an s orbital in principal quantum numbers two and three respectively. Analogously, 2p orbitals are those of principal quantum number two (2px, 2py or 2pz).
In the same way, we can also specify an electron in a particular orbital. Thus a 1s electron or a 3d electron means an electron in the s orbital of principal quantum number one or an electron in the d orbital of principal quantum number three, and so on.
Electronic Structures
Electrons are added to each atom, one at a time across a row. The lowest energy shell fills first then the next and so on. Within a shell, The lowest energy orbitals fill first and once those are full, the next lower energy ones fill and so on.
Structures which show the n-values and their orbitals that are occupied, together with the number of electrons in them are called electronic structures. The question we can ask at this point is: ''how many electrons can a single orbital accommodate?''.
The answer to this question was arrived at by Wolfgang Pauli, who introduced a restriction on the number of electrons which can go into a single orbital. The restriction called the Pauli Exclusion Principle states that ''any orbital will not hold more than two electrons''. This means that any orbital can hold 0, 1 or 2 electrons but not more than 2.
Therefore, the maximum number of electron allowed for each n value will be equal to 2n2 (see Table 1.2). When one electron occupies an orbital, it is said to be unpaired, when there are two electrons in an orbital, they are said to be paired.
Remember, in any atom the K shell is the lowest in energy followed by the L and then the M and so on. Within a shell, the s orbital is the lowest in energy followed by the p orbitals and then the d orbitals and so on.
The lowest energy shell and the lowest energy orbital in that shell fills first. With this and the Pauli Exclusion Principle in mind, we can now write the electronic structures for some of the atoms (Note: In order to write electronic structures of atoms you will need to refer to the periodic table of elements. One is included at the back of this module).
We begin with the simplest atom, hydrogen. This atom has got only one electron, and this will therefore, go into the s orbital of the K shell. The electronic structure we can write for hydrogen is therefore, 1s1.

The lowest energy level of an atom is called the ground state of that atom. For example, we have seen above that in hydrogen the electron normally resides in the 1s orbital.
The electronic state 1s1 is the lowest energy state for hydrogen and it is therefore the ground state. However, the electron in hydrogen can also move to higher levels, say to a 2s orbital. Such states of higher energy are called excited states of the atom.
Note that the s orbital of the K shell of hydrogen is half-full so that in the next atom, helium, an added electron will go into the same orbital and the electronic structure for helium is therefore 1s2.
At this point, the s orbital of the K shell is full in accord with Pauli Exclusion Principle. In the next atom, lithium (Li), the L shell starts filling and the electron will go into the s orbital of this shell (n = 2). The electronic structure of lithium is therefore, 1s22s1 and that of beryllium (Be) is 1s22s2.
The s orbital of the L shell is now filled up, and in the next atom boron (B), the p orbitals of the L shell start filling so that the electronic structure of boron is 1s22s22p1. Since there are three p orbitals, six electrons are required to fill them completely and the atoms B, C, C, O, F and Ne all fill up the 2p orbitals with Ne having the electronic structure 1s22s22p6.
The trend is the same in the third row of the periodic table. The s orbital fills first for this row (Note: this is the s orbital of the M shell) in Na and Mg, followed by the p orbitals. The d orbitals are not filled. In the fourth row, the s orbital of the N shell fills first (in K and Ca). In the next atom scandium (Sc), the d orbitals of row three become lower in energy than the p orbitals of row four, and so they start filling.
The electronic structure we can write for scandium is therefore, 1s22s22p63s23p64s23d1. The d orbitals require ten electrons (for the five of them) and the ten atoms Sc to Zn fill these orbitals. After the 3d orbitals are filled, the p orbitals of the fourth row start filling. Again, note that the d and f orbitals of the fourth row are not filled at this point.
The d orbitals for row four are filled in the fifth row after the s orbital of that row is filled. In the sixth row, the s orbital of that row fills first and then the next electron in La (atom 57) enters the 5d orbital after which, the f orbitals of row four (elements Ce-Lu) start filling.
These require fourteen electrons (for the seven of them) and so we see a group of fourteen atoms normally written at the bottom of the table. After the 4f orbitals are filled, the 5d orbitals continue filling in the remaining nine atoms (Hf-Hg; note that one 5d electron went into La) followed by the 6p orbitals.
The filling up of electrons into orbitals becomes complicated as we go down the table so that we will not continue any further. From the foregoing discussion, we note that the orbitals are filled in the order:
1s<2s<2p<3s<3p<4s3d<4p<5s<4d<5p<6s<4f<5d<6p....
The electronic structures are also written in the same order. There are three points you need to note in these structures. The first is that the sum of the superscripts in the electronic structure gives the atomic number of the atom.
For example, in the electronic structure of oxygen, 1s22s22p4, the sum of the superscripts is 8 (2+2+4), so the atomic number of oxygen is 8. This means that once you know the atomic number of an atom, you can easily write its electronic structure and vice versa.
The second is that the sum of the electrons in the highest principal quantum number, especially for the main group atoms, gives the group number of that atom. For example, in the electronic structure of oxygen, the highest principal quantum number is 2, and there are 6 electrons there (2 + 4 = 6). So oxygen is a group 6A element. Finally, the highest principal quantum number in the electronic structure gives the row (period) in which that atom appears.
In the example of oxygen, the highest principal quantum number in its electronic structure is 2, so oxygen is in row or period 2.
Hund's rule
The Pauli Exclusion principle stated earlier tells us exactly how many electrons can go into a particular orbital; an orbital will not have more than two electrons. With this principle, we can straight away write the electronic structure for example, carbon as, 1s22s22p2. In Section above, we saw that there are three degenerate p orbitals designated npx, npy, npz.
The question now arises: "how do the two 2p electrons of carbon enter the three p orbitals?". There are two possibilities; either both can go into the 2px orbital (i.e., 1s22s22px2, with the 2py and 2pz orbitals empty) or one can go into the 2px and the other one into the 2py (i.e., 1s22s22px12py1 with the 2pz orbital empty).
Friedrich Hund came up with a rule that is used to decide which electronic structure will be the most stable. The rule, called Hund's rule states that "electrons will occupy degenerate orbitals singly if those orbitals are empty".
This means that as long as there is a set of orbitals with the same energy, the electrons will go in separate orbitals until each such orbital has one electron. After that, additional electrons will start pairing up in accord with the Pauli Principle. With this, we can write the most stable electronic structure of carbon as: 1s22s22px12py1 (note: the choice of which orbital is occupied i.e., whether the 2px, 2py or 2pz, is purely arbitrary).
The electronic structures of nitrogen, oxygen, fluorine and neon are therefore: N: 1s22s22px12py12pz1, O: 1s22s22px22py12pz1, F: 1s22s22px22py22pz1, Ne:1s22s22px22py22pz2. Note that nitrogen has one electron in each of the p orbitals in accord with Hund's rule and that pairing starts in oxygen. The same trend applies to the d orbitals and also, the f orbitals.
Paramagnetism and diamagnetism
The electronic structure of carbon given above has two unpaired electrons in the p orbitals while nitrogen has three such electrons. Oxygen and fluorine have two and one unpaired electrons respectively, while neon has no unpaired electrons.
Elements such as carbon, with unpaired electrons, tend to be attracted by a magnet and are said to be paramagnetic. Elements such as neon with all the electrons paired tend to be repelled by a magnet and are said to be diamagnetic.
10 Effects of Being Lazy All the Time

10 Effects of Being Lazy All the Time
Do you find yourself constantly putting things off, feeling unmotivated, and struggling to stick to a routine? If so, you might be dealing with the effects of laziness. While it may seem harmless at first glance, being lazy all the time can actually have numerous negative impacts on your physical health, mental wellbeing, and social life.
In this blog post, we'll explore 10 different ways that being lazy can affect your life – from weight gain and depression to social isolation and lost interests. So let's dive in and discover how laziness can impact every aspect of your life!
10 Effects of Being Lazy All the Time
1. It's Hard to Get Motivated When You're Lazy All the Time
When you're feeling lazy, it can be incredibly difficult to find the motivation to do anything. You might have a long list of tasks that need to be completed, but simply cannot bring yourself to start working on them. This lack of motivation can quickly spiral out of control and lead to feelings of frustration and helplessness.
One reason why it's so hard to get motivated when you're lazy is because your brain becomes accustomed to inactivity. When you spend most of your time sitting around doing nothing, your brain starts to believe that this is the norm. As a result, it becomes increasingly difficult for you to motivate yourself into action.
Another factor that contributes to laziness-induced demotivation is low energy levels. When you don't get enough physical exercise or proper nutrition, your body doesn't produce enough energy for you to feel alert and focused throughout the day. This makes it much harder for you to summon up the drive needed for productivity.
When procrastination becomes routine due being too lazy frequently., its negative effects become ingrained in our habits making us more prone towards procrastination as well as increases anxiety regarding work-related issues leading people towards even lazier behavior than before!
2. You Tend to Procrastinate When You're Feeling Lazy
Procrastination can be a common side effect of laziness. When you're feeling unmotivated and lazy, it's easy to put off tasks that require effort or energy. It's much easier to stay in bed all day than it is to get up and tackle your responsibilities.
However, procrastination often leads to more stress and anxiety in the long run. The longer you put things off, the more overwhelming they become when you finally have to face them. This cycle can be difficult to break out of, especially if you're struggling with chronic laziness.
One way to combat procrastination is by setting small goals for yourself each day. Start with something simple like making your bed or going for a short walk outside. As you accomplish these small tasks, you'll start building momentum towards tackling bigger projects.
Another helpful tactic is breaking down larger tasks into smaller ones. This makes the task feel less intimidating and helps prevent overwhelm from setting in.
It's also important to address any underlying issues that may be contributing to your laziness and procrastination habits. Are there certain triggers that cause your lack of motivation? Are there changes you can make in your lifestyle or environment that might help?
Ultimately, overcoming procrastination takes time and effort but taking small steps towards progress each day will lead towards success over time!
3. Being Lazy Can Lead to Weight Gain and Laziness
Being lazy all the time can lead to a sedentary lifestyle, which in turn can lead to weight gain. When you're not moving around and burning calories, it's easy for excess fat to accumulate in your body. Moreover, being lazy means that you're less likely to engage in physical activities like going for walks or hitting the gym.
In addition to gaining weight, being lazy can also make you feel more lethargic and sluggish throughout the day. You may find yourself lacking energy and motivation even for simple tasks like getting out of bed or doing household chores. This is because when we don't engage our bodies regularly with exercise or movement, our overall physical health suffers.
Furthermore, laziness often leads to poor eating habits as well. When we're feeling too unmotivated or lazy to prepare healthy meals at home, we tend to indulge in fast food and junk food instead. These foods are high in calories and low in nutrients - a recipe for unhealthy weight gain.
To combat these negative effects of laziness on our physical health, it's important that we make an effort towards regular exercise and healthy eating habits. Even small changes such as taking a short walk every day or swapping out sugary snacks for healthier options can make a big difference over time!
4. It's Difficult to Stick to a Routine When You're Lazy
Do you struggle with sticking to a routine? Are you someone who constantly hits the snooze button on your alarm, skips morning workouts and procrastinates important tasks? If so, then laziness could be the culprit.
When we are lazy all the time, it becomes difficult to establish a routine. We may lack the motivation and energy needed to stick to a consistent schedule. Waking up early or completing daily responsibilities can feel like an overwhelming burden when laziness takes over.
Our inability to stick to a routine can have negative consequences on our productivity and overall well-being. Without structure in our daily lives, we may fall behind on work assignments or neglect self-care practices such as exercise or healthy eating habits.
However, there are ways that we can combat laziness and establish routines that work for us. It starts with setting small goals and prioritizing tasks based on importance. Creating a schedule or using productivity tools such as timers or calendars can also help keep us accountable.
While it may seem challenging at first, establishing a routine can lead to increased focus and efficiency in our daily lives. Taking small steps towards overcoming laziness is essential for achieving long-term success both personally and professionally.
5. You May Start to Feel Depressed When You're Lazy All the Time
When you're always feeling lazy, it's easy to fall into a pattern of lethargy that can have negative consequences for your mental health. One of the most common effects of being lazy all the time is depression.
Depression can be caused by many factors, but one major contributor is a lack of physical activity and motivation. When you're not pushing yourself to accomplish tasks or engage in activities that bring you joy, it's natural to feel down and unmotivated.
Additionally, being lazy often leads to social isolation, which can also contribute to feelings of loneliness and depression. When we don't interact with others regularly or participate in social events, our mood may suffer as a result.
Moreover, when we are constantly putting off important tasks due to laziness, it can lead to feelings of guilt and worthlessness - both major contributors to depression. This vicious cycle can be difficult to break out of without taking action.
It's important not only for our physical health but also for our mental well-being that we make an effort every day towards staying active and motivated. Even small steps like going for a walk or tackling one task on your checklist helps combat those feelings of lethargy and prevent them from snowballing into something more serious like depression.
6. Being Lazy Can Lead to Social Isolation
Being lazy can have a profound impact on your social life. When you're feeling unmotivated, you may not want to go out and spend time with friends or family. This can lead to social isolation, which can exacerbate feelings of laziness and depression.
When you isolate yourself from others, it becomes easier to fall into the trap of laziness. Without anyone around to hold you accountable or motivate you, it's easy to slip further and further into a state of inactivity.
Furthermore, being isolated from others can make it difficult to maintain relationships. People may stop inviting you out because they know that you'll decline or cancel at the last minute due to laziness. Over time, this pattern can damage friendships and strain familial ties.
Social isolation can also impact your mental health. Humans are social creatures by nature - we thrive on connection and interaction with others. When we don't get enough stimulation in these areas, our mental health can suffer as a result.
Being lazy doesn't just affect your productivity - it has far-reaching consequences for your entire life. By recognizing how laziness impacts different areas of your life (including socially), you'll be better equipped to address the root causes of this behavior pattern and start making positive changes for yourself moving forward.
7. You May Lose Interest in Things That You Used to
Being lazy all the time can lead to a lack of interest in activities that you once enjoyed. It's important to note that this isn't always a sudden change, but rather a gradual decline. At first, it may simply feel like you're too tired or unmotivated to engage in your favorite hobbies or spend time with friends.
Over time, however, this disinterest can become more pronounced and it may become difficult to muster up any enthusiasm for things that used to bring joy and fulfillment. This could be anything from playing sports or practicing an instrument, to socializing with loved ones.
The loss of interest in these activities can have far-reaching effects on your overall well-being. Without the enjoyment and sense of purpose that comes from engaging in meaningful pastimes, life can start to feel dull and monotonous.
It's important not to beat yourself up over this shift in interests; instead try exploring new hobbies or trying out different experiences that you think might pique your curiosity. By being open-minded and willing to step outside of your comfort zone, you might just discover something new that reignites your passion for life again!
8. It's Hard to Get out of Bed in the Morning
We all know that getting out of bed in the morning can be a struggle, especially if you're used to sleeping in late. But did you know that being lazy and staying in bed all day can actually make it even harder to get up on time?
When we sleep too much or stay in bed for extended periods of time, our bodies become accustomed to this routine and it becomes increasingly difficult to break the habit. This is because our circadian rhythms become disrupted, which affects our ability to fall asleep at night and wake up feeling refreshed in the morning.
Furthermore, staying in bed for excessive amounts of time has been linked to depression and anxiety. These mental health issues can make it even more challenging to find motivation to get up and start your day.
But don't worry - there are ways you can train yourself to wake up earlier and feel more energized throughout the day. Some tips include setting a consistent sleep schedule, avoiding caffeine later in the day, and exposing yourself to natural light as soon as possible after waking up.
So next time you're tempted to hit snooze on your alarm clock, remember how important it is for both your physical and mental health to kickstart your day with some activity!
9. You're Always Making Excuses
Making excuses is a common behavior of lazy people. They always find reasons to postpone or avoid doing something that needs to be done. Excuses are like barriers between you and your goals. They keep you from taking action and achieving success.
One common excuse is "I'm too tired." Lazy people often use fatigue as an excuse for not doing anything productive, even if they have had plenty of rest. The truth is, laziness can make you feel more tired because you're not getting enough exercise or doing activities that energize you.
Another popular excuse is "I don't have time." This excuse is often used by lazy people who spend hours on social media or watching TV instead of focusing on their responsibilities. Time management skills can help overcome this issue.
"I'll do it later" is another common phrase uttered by the chronically lazy person. Procrastination only leads to stress and anxiety in the long run, making tasks seem much bigger than they really are.
Making excuses only perpetuates laziness and keeps individuals from reaching their potential. Avoiding responsibility through justifications will never lead to personal growth nor progress towards goals set forth in life. It's important to identify these tendencies within ourselves so we can actively work against them rather than let our natural inclination towards ease control us indefinitely.
10. You're Always Getting Sick
We all know that being lazy can have a negative impact on our physical health. One of the most common effects of laziness is getting sick frequently. This happens because when we are lazy, we tend to ignore healthy habits such as exercising and eating well.
When you're constantly lounging around instead of engaging in physical activity, your body becomes weak and vulnerable to illnesses. In addition, laziness often leads to poor hygiene practices like skipping showers or neglecting basic self-care routines which can increase the risk of infection and diseases.
Furthermore, people who are always lazy may also experience increased stress levels due to procrastination and missed deadlines. Stress is known for weakening the immune system, making it harder for our bodies to fight off infections.
It's important to remember that leading an active lifestyle doesn't mean hitting the gym every day - it could be as simple as taking a daily walk or doing some light stretching at home. By incorporating small changes into your routine, you'll not only feel better physically but also mentally!
Being lazy can certainly take a toll on our health by making us more susceptible to illness and disease. It's crucial that we make an effort towards cultivating healthier habits so that we don't compromise our wellbeing in the long run!
In conclusion, being lazy all the time can have serious effects on your physical and mental health, as well as your productivity and success in life. It's important to recognize the signs of laziness and take steps to overcome them before they become habits that are hard to break.
By setting goals, developing good habits, staying active both physically and mentally, and avoiding procrastination at all costs, you can conquer laziness once and for all. Remember that it's never too late to make positive changes in your life - start taking action today!
Sugarcane Cultivation Farming Techniques For Successful Production

Sugarcane Cultivation Farming Techniques
Sugarcane is a perennial grass which has a potential to grow to a height of 4.3 meters and has a thickness of around 5.1 centimeters and is generally grown in tropical regions. Sugarcane is able to store Carbohydrates in the form of sucrose and this makes it to be a perennial grass of value.
Its economic value comes from stalk sucrose or Carbohydrates, which it reserves and can be grown both in small scale and on large scale special for commercial purposes. The sugarcane is used as a raw material in production of sugar which is used in different countries. Most of world's sugar we use is made from sugar cane, making it a hot business.
The success of sugarcane production, depends on farmers agronomic skills on can production. This article will provide in detail on how to grow sugarcane both on a small scale and commercial large scale production.
Climatic Requirements
Temperature
Sugarcane performs well in warm temperate areas and is mostly grown in tropical and subtropical areas. The optimum temperature for cultivating sugarcane is known to be in a range of 20 to 35 °C and during germination it requires an optimal range of 32 to 38 °C.
Sugarcane plant growth and maturation depend much on weather conditions on top of good agronomic practices. Sugarcane performs well in frost free areas characterized by warm and sunny. High humidity accelerates cane elongation.
Rainfall
For successful growth, sugarcane requires an annual rainfall range of 1,500 to 2,500mm and any moisture shortfall, should be supplied with Irrigation. The minimum annual rainfall requirement is known to be 600mm and it can be grown successfully in tropical and subtropical climate regions.
Soil requirements
Sugarcane performs well on sandy loam soils which has good soil drainage. The optimum soil pH for growing sugarcane is known to be in a range of 6 to 7.6. Make sure the soil has right moisture content and keep on checking to avoid waterlogging which might be caused through Irrigation.
Cultivation Practices
Propagation
Sugarcane cultivars are propagated for successful production. You stem cut 2-3 lateral buds from immature canes (known as setts). After planting the setts the buds turn into primary and secondary stalk.
The average cane planting density is known to be in a range of 15,000 to 24,000 cuttings per hectare. Setts can be in 30 to 45 centimeters in length and can be planted to a depth of 15 to 30 centimeters. You can grow your setts in nursery and later transplant them to actual field and this will provide easy attention to the setts.
Seed cane
Even though sugarcane can be propagated vegetatively, it doesn't necessarily mean it cannot be grown from seeds. Make sure your field is set and plant your cane free seeds. A 2.5 kg of cane seeds is enough for a hectare.
Seed treatment
Make sure you immerse your cane seeds in hot water having a temperature around 51 °C for 2 hours to soften the seeds for successful germination.
Soil preparation
Avoid growing your sugarcane continuously on the same piece of land, make sure you allow it to fallow. After fallowing, make sure the land is ploughed thoroughly to a depth of 50 to 75 cm. This can be best achieved through the use of a tractor, however, it can also be achieved manually by using any farm tool, i.e. hoe.
Planting
The cane can be planted manually or mechanically, all what you need to do is to choose either of these which is cost effective. Make sure you place your setts in furrow together with a fertilizer for optimal growth, then you can cover with a soil and water it. Make sure setts are planted 100 cm deep.
Planting time
For optimal yield, make sure you plant your cane under Irrigation in autumn weather. In rainfall, it can be planted in spring.
Row spacing
The recommended row and plant spacing for sugarcane production for optimal growth under manual planting is known to be 1.0 to 1.3 m x 0.5 m, while for mechanical is known to be 1.4 and 1.6 m. The setts are planted in a slated form (at a none 90 ° angle).
Fertilization
Sugarcane requires application of appropriate fertilizers and the following can be applied to your cane field per hectare; 80 kg of urea and 100 kg of Single Super Phosphate. All these should be applied in the form of a spray. Decomposing fertilizer can also be applied.
Irrigation
Sugarcane plants require more water application, so make sure you supplement additional water through Irrigation but make sure the site is kept on check from waterlogging, similarly from moisture stress.
Weed Control
Always make sure your cane field is free from weeds as these can be sources of pests and diseases and also they can be competing soil nutrients with your cane plants. Successful weeding can be done manually or by use of herbicides.
Harvesting
Make sure you harvest your cane when it has reached a height of 2 to 4.1 m and usually this is achieved in 12 to 16 months from the time planting is done. Harvesting can be done manually by using a knife and this is applicable to a small cane field, however, on large scale production, make sure you harvest your cane mechanically to make it more efficient.
15 Signs Which Shows You are Pregnant

15 Signs Which Shows You are Pregnant
Are you experiencing unusual symptoms and wondering if you're pregnant? Whether it's your first time or not, the suspense can be overwhelming. But don't worry! We've got you covered with this comprehensive list of 15 signs that indicate pregnancy.
From morning sickness to cravings, we'll help decode those mysterious signals your body is sending out so that you can take action accordingly. So let's dive in and find out if there's a little bundle of joy on the way!
15 Signs Which Shows You are Pregnant
1. Missed Period
A missed menstrual cycle is typically viewed as a reliable indication that someone may be pregnant. However, there are other potential reasons for why you might have skipped your period, such as stress, illness, or disruption to your regular routine. If you think there's a chance you could be pregnant, the best thing you can do is take a pregnancy test.
There are numerous options available to confirm pregnancy, from urine or blood tests. Your doctor can advise you on the best choice for your situation. After getting positive results on your pregnancy test, it's time to start thinking about next steps for caring for your pregnancy. Your doctor can provide support and offer guidance in finding helpful resources for your pregnancy.
2. Morning Sickness
There are many signs that show a woman is pregnant, but one of the most common and well-known signs is morning sickness. Morning sickness is when a woman feels nauseous and sometimes vomits in the morning.
It is caused by the changes in hormones that occur during pregnancy. Morning sickness usually starts around the sixth week of pregnancy and goes away by the 12th to 14th week. Some women have mild morning sickness, while others have severe morning sickness that requires them to be hospitalized.
3. Fatigue
If you're feeling unusually tired lately, it may be a sign of pregnancy. This fatigue is typically experienced at the time of implantation and can be due to shifting blood sugar levels or hormones. You could find yourself requiring more naps during the day or going to bed earlier than normal.
4. Food Cravings or Aversions
If you're experiencing food cravings or aversions, it could be a sign that you're pregnant. Some women crave specific foods during pregnancy, while others find that they can't stand the sight or smell of certain foods. If you suddenly can't stomach your morning coffee or you're dreaming about a particular food, it could be a sign that you're pregnant.
5. Bloating
Many women experience bloating as one of the earliest signs of pregnancy. This is likely due to rising levels of progesterone, which slows down the digestive process and causes gas accumulation in the stomach and intestines. For some women, this can be their first indication that they are expecting.
6. Headaches
If you're experiencing headaches during your pregnancy, there could be a variety of causes. Changes in hormones can result in your blood vessels dilating and this can lead to headaches. Lack of sleep, dehydration and hunger are also potential reasons for pregnancy-related head pain.
To ease your discomfort, aim to stay hydrated, get the rest you need and if it doesn't helps, apply a cold compress to your forehead. It is best to consult with your doctor if the intensity or frequency of headaches persists.
7. Constipation or Diarrhea
If you are pregnant, constipation and diarrhea can be common side effects. Constipation is usually brought on by the rise of hormones in your body, which makes it hard to have a bowel movement as they may be slowed down. Your stools might become harder and drier too.
Diarrhea can also occur as a result of the increased hormones or other possible factors like an infection or food poisoning. To avoid dehydration, make sure to stay hydrated when having this symptom.
7. Increased Urination
Frequent urination can be an indication of pregnancy. The additional blood in your body increases the workload of your kidneys and you produce more urine as a result. Simultaneously, the expanding uterus exerts strain on your bladder, prompting you to need to go more often. Pregnant women may additionally experience having to wake up several times during the night for bathroom trips.
8. Changes in Skin and Hair
Your body may be undergoing changes as it readies itself to nurture a growing embryo. Changes in your skin and hair may be due to a surge of hormones, possibly causing your skin to feel oilier or drier than usual and your hair being shinier or coming out more. Some pregnant women also get dark patches called melasma on their face. Generally speaking, these changes should not cause alarm, but talk to your doctor if you have any concerns about the changes in your skin or hair.
9. Swelling of the Breasts
Pregnancy often causes the breasts to swell, typically during the earliest stages. This is due to heightened hormone levels in the body. Tenderness, soreness and a feeling of fullness may result, along with darkening or enlargement of the area around the nipple, known as the areola.
10. Mood Swings
If you're experiencing mood swings during pregnancy, it's normal. You may feel elated one minute and down the next. These sudden changes in mood are due to hormonal fluctuations. As your pregnancy progresses, your hormone levels will continue to fluctuate, which can cause mood swings. If you're feeling particularly anxious or stressed, talk to your healthcare provider about ways to manage your emotions.
11. Increased Sense of Smell
If you're pregnant, your sense of smell might become more acute than usual. This increased sensitivity to odors, called hyperosmia, is often a symptom of pregnancy. Foot high levels of hormones can lead to other signs such as nausea and vomiting (morning sickness), as well as a heightened reaction to smells.
You may find that some scents are especially overpowering or negatively affect your mood. To be sure, it's essential to consult with a doctor who can determine whether or not you are expecting - as there are other reasons why someone may have hyperosmia.
12. Pregnancy Glow
As your pregnancy progresses, you may notice that your skin is changing. You may have a “glow” that comes from increased blood flow and the hormones of pregnancy. This can make your skin look and feel softer, smoother, and brighter. Your hair may also be growing faster and appearing shinier than usual.
13. Hives or Other Skin Rashes
If you are suddenly stricken with hives or any other kind of skin rash, it may be a sign that you are pregnant. This is because your immunity tends to be diminished during pregnancy, leaving you more prone to skin reactions. Therefore, if you are experiencing additional signs of pregnancy like fatigue and nausea alongside the rash, it would be wise to do a pregnancy test and then see a doctor for confirmation.
14. Weight Gain or Loss
Pregnancy can show several signs; weight gain or loss being one of them. Most women tend to experience increased appetite and water retention due to the higher hormone levels, leading to weight gain during the first trimester. Then again, some have reported weight loss due to morning sickness. If you are pregnant, it is advisable to consult with your doctor regarding your expected body mass changes throughout pregnancy.
15. Darkening of the Areolas
As your body prepares for breastfeeding, the Montgomery glands on your areolas (the dark circles around your nipples) enlarge and secrete an oily substance. This can make your areolas appear darker than usual.
The Future of Artificial Intelligence: Trends and Predictions
The Future of Artificial Intelligence: Trends and Predictions
Artificial Intelligence (AI) is no longer just a futuristic concept found in sci-fi novels. It's becoming an integral part of our daily lives, shaping industries and transforming how we interact with technology. From virtual assistants like Siri and Alexa to advanced algorithms that power self-driving cars, AI has seeped into various facets of society.
But what does the future hold for this rapidly evolving field? As advancements continue at breakneck speeds, understanding the trends and predictions surrounding AI becomes essential. This exploration opens doors not only to exciting innovations but also raises critical questions about ethics and societal impact. Let’s dive deeper into the current landscape of AI and uncover what lies ahead on this thrilling technological journey.
Understanding Artificial Intelligence (AI)
Artificial Intelligence encompasses a range of technologies designed to perform tasks that typically require human intelligence. This includes problem-solving, learning, and even understanding language.
At its core, AI relies on data. Machines analyze vast amounts of information to recognize patterns and make decisions. The more data they process, the smarter they become.
There are two main categories: narrow AI and general AI. Narrow AI excels in specific tasks like facial recognition or language translation. General AI, still largely theoretical, aims for machines with human-like cognitive abilities.
Machine learning is a significant subset of AI where algorithms improve through experience without explicit programming. It's this capability that enables applications ranging from recommendation systems to predictive analytics.
Understanding these fundamentals lays the groundwork for exploring how artificial intelligence can revolutionize various sectors in ways we’re only beginning to imagine.
Current Applications of AI
Artificial Intelligence is transforming the way we interact with technology daily. In healthcare, AI algorithms analyze medical data to detect diseases earlier than traditional methods. This leads to more effective treatments and improved patient outcomes.
In finance, AI-driven tools assess risks in real-time. They help banks prevent fraud and streamline loan approvals, making financial transactions faster and safer.
Retail has also embraced AI through personalized recommendations. Online shopping platforms use sophisticated algorithms to suggest products based on customer behavior, enhancing user experience.
Moreover, AI chatbots are revolutionizing customer service by providing instant support around the clock. Businesses can resolve inquiries quickly while improving client satisfaction.
Even in agriculture, farmers utilize AI for precision farming techniques that optimize crop yields while minimizing resource use. These applications illustrate just a fraction of how deeply integrated artificial intelligence has become across various sectors today.
The Impact of AI on Different Industries
Artificial Intelligence is reshaping various industries, driving efficiency and innovation. In healthcare, AI algorithms analyze medical data to improve diagnosis and treatment plans. This technology accelerates drug discovery and personalizes patient care.
In finance, machine learning models assess risk more accurately than traditional methods. Automated trading systems can make split-second decisions for investors, optimizing their portfolios in real-time.
The retail sector benefits from AI through personalized shopping experiences. Algorithms recommend products based on consumer behavior, enhancing customer satisfaction.
Manufacturing sees significant advancements as well. Robotics powered by AI streamline production processes while minimizing errors. Predictive maintenance reduces downtime by anticipating equipment failures before they occur.
Education also embraces AI with personalized learning tools that adapt to students’ needs. Intelligent tutoring systems provide tailored feedback that promotes better understanding of subjects.
Each industry faces unique transformations driven by this powerful technology, paving the way for a smarter future.
Ethical Considerations and Concerns
As AI technology evolves, ethical considerations take center stage. The potential for bias in algorithms raises serious questions about fairness and equality. If not properly managed, AI can perpetuate or even exacerbate existing inequalities.
Privacy is another pressing concern. With vast amounts of data collected to train these systems, individuals often find their personal information at risk. Striking a balance between innovation and protecting user rights becomes essential.
Moreover, accountability remains a gray area. Who takes responsibility when an AI system makes a mistake? These challenges require ongoing dialogue among technologists, ethicists, and policymakers.
Transparency in AI decision-making processes is also vital. Users must understand how decisions are made to trust the technologies that impact their lives daily.
Navigating these ethical waters demands cautious deliberation as we move forward with artificial intelligence integration into society.
The Future of AI: Predictions and Possibilities
The future of AI is a landscape filled with endless possibilities. As technology evolves, we can expect more sophisticated algorithms that learn and adapt at unprecedented speeds.
One prediction suggests that AI will enhance human capabilities rather than replace them. This collaboration could lead to breakthroughs in various fields, from healthcare to finance. Imagine doctors using AI to analyze patient data for faster diagnoses or financial analysts predicting market trends with greater accuracy.
Another possibility lies in the realm of creativity. We may see AI generating art, music, and literature that provokes thought and emotion, blurring the lines between human and machine-generated content.
However, as these advancements unfold, questions about accountability arise. Who takes responsibility for decisions made by intelligent systems?
Navigating this new terrain will require careful consideration of both innovation and ethics as we embrace what’s next on the horizon for artificial intelligence.
Advancements in AI Technology
The landscape of AI technology is evolving rapidly. Breakthroughs in deep learning and natural language processing are reshaping how machines interact with humans.
New algorithms enable AI to understand context better than ever before. This enhances everything from virtual assistants to customer service bots. They can now provide more accurate responses based on user intent.
Computer vision also sees remarkable progress. Machines can analyze images and recognize patterns with impressive accuracy. Applications range from healthcare diagnostics to autonomous vehicles, pushing the boundaries of what’s possible.
Quantum computing is on the horizon too, promising a leap in processing power for complex AI tasks. This could revolutionize fields like drug discovery and materials science.
As these advancements unfold, they will unlock new capabilities, making AI an even more integral part of our lives. The possibilities seem endless as we continue to innovate and explore uncharted territories in artificial intelligence technology.
Potential Benefits and Risks of Advancements in AI
Advancements in AI hold immense potential. They can enhance productivity across various sectors, streamline processes, and drive innovation. From personalized healthcare solutions to automated customer service, the possibilities are vast.
However, these advancements also come with risks. Job displacement is a significant concern as machines take over tasks traditionally performed by humans. This shift could lead to economic disparities if not managed properly.
Moreover, bias in AI algorithms poses challenges for fairness and equality. If not carefully monitored, technology could perpetuate existing societal biases.
Data privacy is another pressing issue. As AI systems become more integrated into daily life, the risk of unauthorized data access increases.
Navigating this landscape requires balance—leveraging benefits while mitigating risks effectively. Awareness and proactive measures will be key as we embrace the future shaped by artificial intelligence.
Embracing the Future of AI Responsibly
As we step into a future shaped by artificial intelligence, responsibility is key. Organizations must prioritize ethical considerations when developing AI systems. This means ensuring transparency in algorithms and data usage.
Collaboration among technologists, ethicists, and policymakers can foster trust. Engaging diverse perspectives helps avoid biases that might arise from limited viewpoints.
Education plays a vital role as well. By equipping individuals with knowledge about AI technologies, society becomes better prepared to navigate the complexities they introduce.
Proactive regulation offers another layer of safety. Establishing guidelines for accountability ensures that AI serves humanity’s best interests without infringing on privacy or autonomy.
Fostering an environment where innovation thrives alongside ethical standards will pave the way for a brighter technological tomorrow. Embracing this balance opens doors to possibilities while safeguarding fundamental values.
Conclusion
The evolution of artificial intelligence is undeniable. As we look ahead, the integration of AI into everyday life will become even more seamless and complex. Innovations in machine learning, natural language processing, and robotics are set to transform various sectors.
However, this rapid advancement comes with its share of challenges. Ethical considerations surrounding privacy and job displacement must be addressed as society embraces these technologies. It’s essential for stakeholders—from developers to policymakers—to collaboratively shape a future where AI serves humanity's best interests.
As we venture further into the era of AI, embracing both its potential benefits and inherent risks will be crucial. The way forward lies in responsible innovation that prioritizes ethical standards while harnessing the transformative power of artificial intelligence. The journey has only just begun; it promises to be an exhilarating ride filled with opportunities for growth and discovery.
Fundamental Quantities and Units
Fundamental Quantities and Units
Introduction
Measurements are part and parcel of everyday life. You may have weighed masses and volume of different substances e.g. 1kg of sugar, 500g of flour, 2litres of water. All substances that you are able to measure are called physical quantities.
In most cases measurements that we take are associated with errors. In actual experiments it is very difficult to record 100% accurate measurements. This is the case due errors that may be associated either with you as a researcher or the instruments being used. All measurements of a physical quantity that you take in science are recorded in SI units.
Physics is a branch of science that deals with measurements. It involves scientific procedures and processes that deal with error analysis in measurements and standard presentation of figures in science. Your understanding of this unit is very important to both Physics and General Science.
Objectives
By the end of this article you should be able to:
- Define physical quantities
- Differentiate derived quantities from basic quantities
- Analyze dimensions of different physical quantities
- Evaluate errors associated physical quantities
Physical Quantities
A physical quantity is a quantity that can be measured. In your homes, schools and in everyday life you take measurements of maize flour, sugar, water, paraffin, etc. All these are examples of physical quantities. We can classify physical quantities in two, scalar quantities and vector quantities.
Scalar Quantities
Quantities that have magnitude only, e.g. mass, temperature, time, length. The magnitude of a scalar quantity indicates its numerical value and unit of measure.
Vector Quantities
Quantities that have both magnitude and direction, e.g. velocity, force, momentum, displacement, etc. The magnitude of a vector is a scalar that indicates only how large or small the vector is.
Units
A quantity used as a standard of measurement is called a unit. In Physics we use two sets of units, thus basic units and derived units.
Basic Units
Basic units are units selected for measuring mass, length and time. These quantities cannot be expressed in terms of other quantities. We have seven basic quantities in physics. The units of all the other physical quantities in both science and engineering are derived from these seven basic quantities.
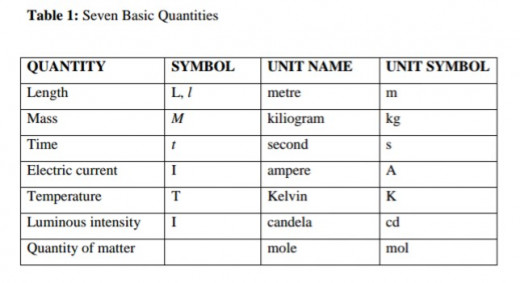
Basic Quantities
Derived Units
Having looked at basic or fundamental units now we want to consider a combination of several basic units. Units obtained from a combination of several basic units through multiplication and division are referred to as derived units. You may alternatively define derived units as units of physical quantities that can be expressed in terms of fundamental units. We normally use units of length, mass and time to come up with derived units.
We have plenty of examples of derived units that we encounter in everyday life for example, velocity, force, work, etc.
Example 1, Show that the unit of force is a derived quantity
Solution:
Velocity = displacement/time= length/time=m/s
Multiples and sub-multiples of SI Units
We can generate multiples and sub-multiples of SI units by adding appropriate prefixes to the units. Every prefix has a numerical value, for instance we use kilo to represent 1000, micro to represents 10-6 , i.e. 20 kilowatts = 20 x 1000 watts = 20 000 watts.
Sometimes prefixes may be represented using a factor or symbol as shown in table 2.
Table 2: Multiples and sub-multiples of SI units

Multiples and Sub-Multiples of SI Units
As you may see from table 2, multiples and submultiples are used to shorten long numbers e.g. we can 20 000 000 000 bites as 20 x 109 = 20gigabites =20 GB.
Conversions
After looking at multiples and sub-multiples, we can now convert SI units from one unit to another.
We use several conversion factors to translate a measurement from one unit to another unit. In table 4, we are translating units from column A to B by multiplying a unit in column B with a factor. For instance to change 1mile into a meter, you have to multiply a meter with a factor of 1609.344.
In cases where a unit cannot be translated directly to other unit, we translate it first to intermediate unit/s and then proceed to find our desired unit.
Table 3: Unit Convertion
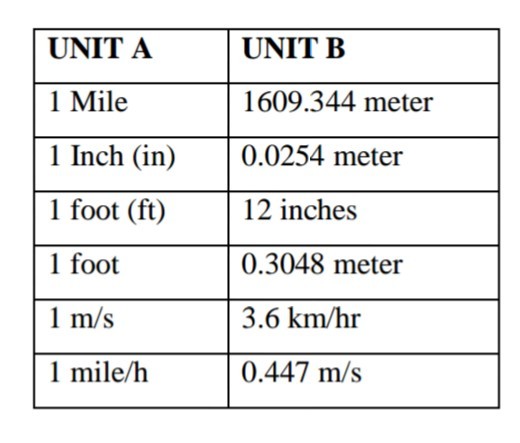
Units Convertion
Dimensions and Dimensional Analysis
A dimension is a physical variable used to specify the behavior or nature of a particular system. E.g. the length of a rod is so many meters.
We use square brackets [ ], to denote dimensions. For example we can represent the dimension of velocity as [V]=[L]/[T]
So far we have defined a dimension, but in most cases we use dimensions to express a dimension. The dimensions of a physical quantity are the powers to which the basic quantities (M, L and T) must be raised to represent a physical quantity. We represent a basic quantity, which does not appear in the physical quantity by raising it to power zero.
Example 2.
Find the dimension of acceleration a, and the dimensions of M, L and T in the dimensional formula of acceleration.
The dimension of acceleration; [a] = [M0LT–1]/[T]= [M0LT–2]
Hence the dimensions of acceleration are 0 in mass, 1 in length and -2 in time. Alternatively we can present dimensions of acceleration as M = 0, L = 1 and T = -2.
Dimensional Analysis
Dimensional Formula
This is the expression of a physical quantity in terms of its dimensions. For example, the dimensional formula for force is
[F]= [MLT–2]
Dimensional Equation
If you express an equation containing physical quantities, each quantity represented by its dimension formula, the resulting equation is referred to as Dimensional Equation.
Consider the formula v= u +at
Here u is initial velocity of the body, a is acceleration and t is the time taken to attain final velocity v. Writing this equation in the dimensional form, we have
[M0LT–1]=[M0LT–1] + [M0LT–1] + [M0L0T]
NOTE: in a dimensional equation, the dimensions in every term should be the same
TABLE 3. Dimensional formulae of Physical Quantities
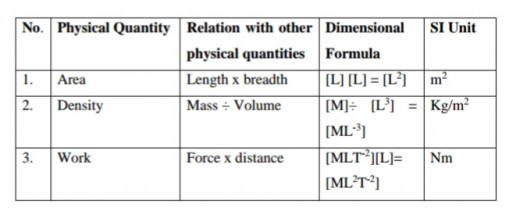
Dimension Formula of Physical Quantities
Categories of Physical Quantities
When you carefully study dimensional formula of the physical quantities you will discover that there are four categories of physical quantities.
a. Dimensional variables: Physical quantities that have dimensions but do not have fixed value e.g. force, velocity, work, power, etc.
b. Dimensional constants: Physical quantities which posses dimensions and have fixed value e.g. gravitational constant, Planck’s constant, etc.
c. Dimensionless variables: Physical quantities, which have neither dimensions nor fixed value e.g. Unfixed Constants: specific gravity, strain,
- Trigonometric functions: Cos 30°
- Logarithmic functions and exponential functions: log 10,
d. Dimensionless constants: Physical quantities, which do not posses dimensions but have a fixed value e.g.
- Numerical values: 1, 2, 200
- Fixed constants: π, index of refraction (n), etc.
Uses of Dimensional Formula (Analysis)
Dimensional analysis is used in the following ways:
- To check the correctness of an equation
- To remember a forgotten formula
- To derive relationship between different physical quantities
- To convert one system of units to another
- To find the dimensions of constants in a given relation
Checking the Correctness of a Physical Relation (Equation)
In order to check if a physical relation or a scientific equation is correct or not, dimensional analysis is applied. When the dimensions of the two sides of the equation thus, the right hand side (RHS) and the left hand side (LHS) are equal a scientific equation is considered as dimensionally correct and visa versa.
Example 3
Show if the equation, v = u+at2 is dimensionally correct or not. Where v is final speed, u is initial speed, a is acceleration and t is time.
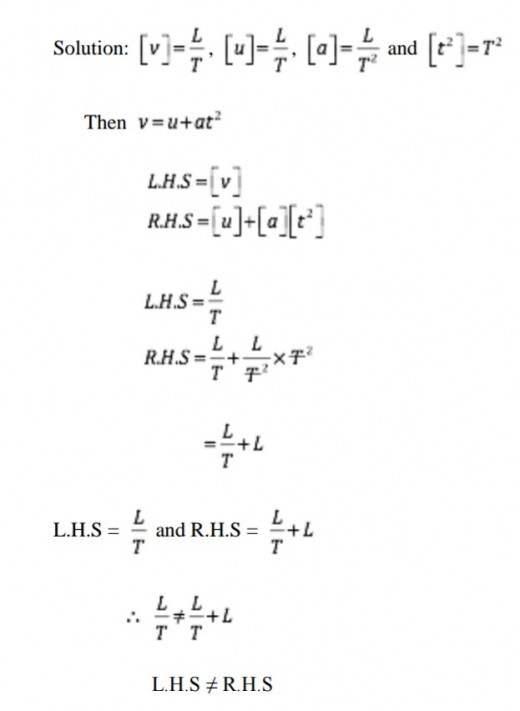
The equation is dimensionally incorrect.
To Calculate Dimensions of Unknown Quantity
When you want to find an unknown variable in an equation, you simply make it the subject of the formula and evaluate dimensions of the terms on the right hand side of the equation.
Example 4
A sphere of radius a, moves with velocity v, in a medium and the force F acting on it, is given as;

As you have observed from example 2, it is easier to work out SI units for an unknown quantity using dimensional analysis. Having found the dimensions of unknown quantity we can proceed to find SI units for unknown quantity. Each dimension gives a unit.
Measurements and Accuracy
Measurements
Measurement is the comparison of unknown physical quantity with a known fixed quantity.
For example if the length of the rod is 3 metres, it means the unit of length is the metre and this unit is contained 3 times in the length of that rod.
Two things represent the magnitude of a physical quantity;
i. The unit in which the quantity is measured e.g. metre
ii. The numerical value – this expresses how many times a unit is contained in a physical quantity, e.g. 3.
Accuracy
You may recall from your secondary school physical science that accuracy is defined as the measure of how close the measured value of a quantity corresponds to its true value. Oftenly we use the term accuracy hand in hand with precision. The degree of reproducibility or the closeness between repeated measurements is called precision.
Every measuring instrument has an inherent limit of precision or accuracy. This limit is determined by the list count of the instruments. No matter how carefully we measure, we can never obtain a result more precise than the limit of our measuring device. In experiments we calculate the limit of precision by considering ½ of the smallest division built into the device.
When you are using a vernier calliper that has a smallest unit of measurement as 0.1mm, its limiting precision is 0.1 x ½ = ± 0.05mm = ± 0.005 cm. Suppose using this vernier calliper, you have measured the length of a rod as 5.95cm, your limit of precision will be the same ± 0.05cm. The length of the lord will be represented as 5.95±0.05 cm. This means the true value of the length lies between 5.95 – 0.05 cm (5.9 cm) and 5.95+0.05 cm (6.0 cm).
Significant figures
The number of digits in a measurement about which we are certain, plus one additional digit which is uncertain are known as significant figures. The greater the number of significant figures obtained in a measurement, the more accurate is the measurement. But in most cases the number of significant figures is limited by a limit of precision.
Rules for significant figures
When we are using significant figures we consider the following rules:
i. All non-zero digits are significant e.g. 157.23 g contains five significant figures
ii. All zeros between two nonzero digits are significant e.g. 305.008 m contains six significant figures
iii. Unless stated otherwise, all zeros to the left of an understood decimal point but to the right of a nonzero digit are not significant e.g. 904,000s contains three significant figures.
iv. All zeros to the left of an expressed decimal point and to the right of a nonzero digit are significant e.g. 406.000 contains six significant figures.
v. All zeros to the right of a decimal point but to the left of a nonzero digit are not significant e.g. 0.000132ml contains three significant figures.
vi. All zeros to the right of a decimal point and to the right of a nonzero digit are significant e.g. 0.08040N and 50.00N each contain four significant figures.
Calculations involving significant figures
You should always remember that mathematical calculations cannot increase either the precision or the number of significant figures of the measured quantities. When we are doing calculations involving significant figures, we apply the following rules:
a. In addition and subtraction, the number of decimal places in the result should be equal to the smallest number of decimal places of any term in the sum or difference of two terms.
b. In multiplication or division, the number of significant figures in the result should be equal to the number of significant figures of the list least precise term in multiplication or division.
Example 5
Evaluate the following:
i. 3.76m+46.855m+0.2m
= 50.815m
Applying the first rule, the position of a decimal place in the result should be the same as in 0.2.
Answer: 50.8
ii. 11.3m x 6.8m
= 76.84m2
Applying the second rule, the number of significant figures in the result will be the same as in 6.8m
Answer: 77m2
Error
An error (inaccuracy) in measurements of any sort is a departure of the output of any measuring system from the value truly representing the quantity being measured.
This departure occurs in practice, no matter how sophisticated the measuring equipment is. We usually define error as the uncertainty in measurement.
Types of errors
1. Systematic errors: These are errors that appear in measurement due to known causes. They include all inaccuracies that tend to be more in one direction than in the opposite one. We can reduce systematic errors by repeating measurements a number of times. Some important causes of such errors are:
- Incorrect design or calibration of the instrument
- Limitation of the method used for the measurement
- Incorrect reading or interpretation of the instrument
- Lack of accuracy of the formula being used
2. Random errors: These are errors which appear in a measurement due to unknown causes. Random errors have no set pattern and their magnitude depends on the researcher’s knowledge of the apparatus used. Taking repeated readings of a quantity and averaging reduces random errors, but not systematic errors.
3. Gross errors: these are errors due to sheer carelessness of the researcher. They mostly involve human errors. Gross errors cannot be corrected but we may reduce them. In most cases we generate gross errors by:
- Recording the observations wrongly
- Reading the instrument incorrectly
- Entering wring values of observations in calculations
- Not caring for sources of error and precision.
Error Analysis
When dealing with errors in physics we oftenly use the following words; true value, absolute error, mean absolute error, relative error and percentage error.
A true value is the arithmetic mean of a large number of readings of a quantity. If during an experiment you have taken a1, a2, a3 ….. an as part of n different readings of a physical quantity in an experiment, then the true value of that quantity is
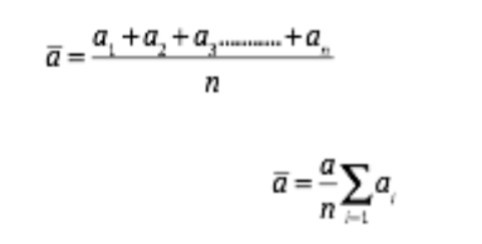
We note from the above equation that the true value is the same as arithmetic mean.
We define absolute error as the difference in the magnitudes of true value and the measured value of a physical quantity. We sometimes refer absolute error to actual error. If you observed a1, a2, a3 ….. an as you n different readings of a physical quantity in an experiment then the absolute errors are given as;


6 Health Benefits of Rabbit Meat

6 Health Benefits of Rabbit Meat
Are you looking for a new source of protein that isn't just delicious but also incredibly healthy? Then look no further than rabbit meat! This lean and versatile meat is packed with essential nutrients and offers numerous health benefits that will leave you feeling satisfied both inside and out. From boosting your immune system to aiding in weight loss, there are plenty of reasons why you should incorporate rabbit meat into your diet.
In this blog post, we'll explore 6 incredible health benefits of rabbit meat that will convince even the most skeptical eater to give it a try. So what are you waiting for? Let's hop right in!
Nutritional Value of Rabbit Meat
Rabbit meat is an excellent source of protein, providing all the essential amino acids your body needs. It's also a lean meat, with very little fat. This makes it a healthy choice for those looking to watch their weight or cholesterol levels. Rabbit meat is also a good source of iron and vitamin B12.
6 Health Benefits of Rabbit Meat
The following are the health benefits of rabbit meat:
1. High Protein Content
Rabbit meat is an ideal source of protein for anyone aiming to up their nutrition. It provides 20-25% protein, far exceeding the amount found in other meats. Getting enough protein is fundamental for muscle growth and energy production - making rabbit an optimal choice.
2. Essential Vitamins and Minerals
Rabbit meat is packed with essential vitamins and minerals, such as vitamin B12, which helps to keep red blood cells and nerve cells healthy. It's also an excellent source of iron that aids in the transportation of oxygen around the body.
Zinc, important for maintaining a strong immune system and aiding topical healing, can also be found in rabbit meat.
3. Low Fat and Cholesterol Content
Rabbit meat is an excellent source of lean protein, meaning that it is low in fat and cholesterol. This makes it an ideal choice for those looking to reduce their risk of heart disease or other health issues related to high levels of fat and cholesterol.
Rabbit meat is also very low in saturated fat, which is the type of fat that can increase cholesterol levels. Additionally, rabbit meat is a good source of monounsaturated fats, which can help reduce cholesterol levels. The low-fat content of rabbit meat makes it an ideal choice for those looking to reduce their fat intake. Eating a diet that is low in fat can help reduce the stroke.
4. Easy to Digest
In addition to its high nutritional value, rabbit meat is also easy to digest. This makes it a great choice for those with digestive issues or food sensitivities.
5. Disease Prevention
Studies have revealed that rabbit meat carries a number of health benefits, such as lowering the chances of developing certain ailments. Evidence has shown that eating rabbit meat can lower the risks of heart problems, stroke, and some types of cancer.
Additionally, its high content of omega-3 fatty acids can reduce inflammation and better your overall health. Rabbit meat is also known to help manage type 2 diabetes. Studies demonstrate that consuming it can regulate blood sugar levels and bolster insulin sensitivity, significantly reducing the risk of developing this medical condition and other metabolic disorders.
6. Rabbit Meat is Good For Pregnancy
Pregnant women need enough rabbit meat to maintain their well-being. This type of meat is packed with essential fatty acids, which are paramount for the mother-to-be and her unborn baby's health. It also provides plenty of protein, required to create new cells and repair any damage related to pregnancy. Additionally, it contains zinc, a nutrient which contributes to the proper development of the infant's nervous system.
The Best Way to Cook Rabbit Meat
Cooking rabbit meat can be done in various ways, however some are more preferable than others. Roasting is one of the best methods you can use to bring out the natural flavors and render it tender. Not only that, but it's a healthy way to prepare it, without any added chemicals or fats. It's important that you make sure the meat is cooked all the way through - check that there is no pinkness before consumption.
Rabbit Meat Recipes
Here are some delicious rabbit meat recipes to try:
- Rabbit Stew: This hearty stew is perfect for a cold winter day. It's made with chunks of rabbit meat, carrots, potatoes, and herbs.
- Rabbit Curry: This flavorful curry is made with diced rabbit meat, coconut milk, and Indian spices. Serve it over rice or noodles.
- Rabbit Stir-Fry: A quick and easy stir-fry that's full of flavor. Made with diced rabbit meat, vegetables, and soy sauce.
- Rabbit Risotto: A creamy risotto made with Arborio rice and chicken broth, flavored with Parmesan cheese and white wine. Diced rabbit meat is added in at the end.
- Roasted Rabbit: A simple recipe for roasted rabbit that results in tender, juicy meat. Season the rabbit with salt and pepper before roasting at a high temperature until browned all over.
In conclusion, rabbit meat is a great source of nourishment that can provide numerous health benefits. Not only does it have high levels of protein, but it’s also rich in vitamins and minerals such as zinc, iron and B12. It also contains essential fatty acids which are good for our heart health and has low fat content compared to other meats. All these make rabbit a great choice for those who want to enjoy delicious meals while improving their overall health.
Personalized Nutrition: How Genetics Impact Your Diet
Personalized Nutrition: How Genetics Impact Your Diet
Have you ever wondered why some diets work wonders for your friends but leave you feeling bloated and unsatisfied? The secret might lie in your genes. Personalized nutrition is revolutionizing the way we think about food, tailoring dietary approaches to fit individual biological makeups.
As more people seek unique solutions to their health and wellness challenges, the connection between genetics and nutrition is gaining momentum. Imagine a diet plan designed just for you—one that takes into account how your body processes nutrients based on its genetic blueprint. Welcome to the fascinating world of personalized nutrition, where understanding your DNA could lead you toward better health outcomes and a happier relationship with food!
Understanding Personalized Nutrition and its Growing Popularity
Personalized nutrition is emerging as a groundbreaking approach in the health and wellness landscape. Unlike traditional dietary guidelines that offer one-size-fits-all solutions, this innovative strategy considers individual variations in metabolism, preferences, and lifestyle.
As people become more health-conscious, they seek tailored solutions that resonate with their unique needs. The rise of wearable technology and mobile apps has empowered individuals to track their nutrition and fitness levels like never before.
Social media plays a key role too. Influencers sharing success stories about personalized diets create buzz around customization. This growing interest fuels research into how our genetics influence our nutritional requirements.
With increasing awareness about the impact of diet on overall health, more individuals are now willing to invest time and resources into understanding what truly works for them. Personalized nutrition offers a fresh perspective—one where each person's journey to wellness can be uniquely crafted.
The Role of Genetics in Determining Individual Nutritional Needs
Genetics plays a pivotal role in how our bodies respond to food. Each of us carries unique genetic markers that influence metabolism, nutrient absorption, and even taste preferences. These biological variations can dictate whether we thrive on a high-carb diet or require more healthy fats for optimal function.
Certain genes are linked to the way we break down macronutrients. For instance, some individuals may have genetic predispositions that make them more sensitive to sugar intake while others might efficiently process it without issue.
Additionally, genetics can impact how well we absorb vitamins and minerals from our diets. This means two people eating identical meals could experience vastly different health outcomes based on their genetic makeup.
Understanding these nuances allows for tailored dietary approaches that accommodate individual needs rather than following one-size-fits-all recommendations. Recognizing your own genetic profile can empower you to make informed choices about what fuels your body best.
Nutrigenomics: The Science Behind Personalized Nutrition
Nutrigenomics is an emerging field that explores the intricate relationship between our genes and nutrition. It delves into how individual genetic variations influence dietary responses and nutritional requirements.
This science examines how certain genes can affect metabolism, nutrient absorption, and even food preferences. For instance, some people may have a genetic predisposition to process fats differently than others.
By understanding these genetic factors, researchers aim to tailor dietary recommendations that align with one’s unique genetic makeup. This personalized approach has the potential to enhance health outcomes significantly.
Through nutrigenomics, individuals can gain insights into which foods will optimize their well-being, prevent diseases, or help manage existing conditions effectively. The promise of this science lies in its ability to transform nutrition from a one-size-fits-all model into a customized experience based on genetics.
How Genetic Testing can Help Customize a Diet Plan
Genetic testing offers a fascinating way to tailor diets specifically for individual needs. By analyzing your DNA, these tests reveal how your body processes nutrients and responds to various foods.
For example, some people may metabolize fats differently than others. This insight allows for a diet plan that emphasizes healthy fats while minimizing those that could lead to weight gain or health issues.
Additionally, genetic markers can indicate sensitivities or intolerances. If you have a predisposition toward lactose intolerance, knowing this in advance helps eliminate unnecessary discomfort when choosing dairy products.
Moreover, genetic insights can guide micronutrient intake. Understanding whether you metabolize certain vitamins efficiently enables the creation of more effective supplementation strategies.
As science advances, personalized nutrition becomes increasingly attainable through simple at-home tests. This approach isn’t just about feeling good; it’s about optimizing health based on what makes you unique.
Case Studies and Success Stories of Individuals Who Have Benefited from Personalized Nutrition
Emily, a 35-year-old fitness enthusiast, struggled with fatigue and digestive issues for years. After undergoing genetic testing, she discovered her body had difficulty processing certain carbohydrates. With this knowledge, she tailored her diet to reduce those specific foods and saw remarkable improvements in her energy levels.
Then there's Jake, an athlete who wanted to optimize his performance. Genetic insights revealed he had a predisposition for lactose intolerance. By switching to dairy alternatives and increasing his protein intake from plant-based sources, he enhanced both recovery times and overall stamina during competitions.
Another inspiring story comes from Maria, who faced weight management challenges throughout her life. Through personalized nutrition based on her genetic profile, she learned which fats benefited her metabolism best. This led to sustainable weight loss without feeling deprived or overwhelmed by restrictive diets.
These stories illustrate the transformative power of understanding one's unique nutritional needs through genetics. Each journey highlights how tailored approaches can lead to remarkable health outcomes.
Challenges and Limitations of Personalized Nutrition
Personalized nutrition holds great promise, but it’s not without its challenges. One major hurdle is the complexity of human genetics. Each person's genetic makeup is unique, making it difficult to create one-size-fits-all dietary recommendations.
Additionally, access to genetic testing can be limited. Not everyone has the resources or desire to undergo such tests, which can lead to disparities in personalized nutrition benefits.
Another concern lies in interpreting genetic data accurately. Misinterpretation can result in misguided diet plans that do more harm than good.
Moreover, while genetics play a role in nutritional needs, environmental factors and lifestyle choices also significantly influence health outcomes. Balancing these aspects requires a multi-faceted approach.
There are ethical considerations surrounding privacy and data security when sharing genetic information for dietary purposes. Addressing these issues is crucial as personalized nutrition continues to evolve.
Future Advancements in the Field of Personalized Nutrition
The future of personalized nutrition is incredibly bright. As technology advances, the integration of artificial intelligence and machine learning into dietary planning is becoming a reality. These tools can analyze vast amounts of genetic data quickly, offering tailored meal plans that align with individual health goals.
Wearable tech will also play a pivotal role. Imagine devices that not only track your activity but also monitor how specific foods impact your body in real-time. This feedback could revolutionize diet management.
Furthermore, collaborations between scientists and food companies are emerging. They aim to create functional foods designed specifically for genetic profiles. Personalized supplements may soon be common as well, providing nutrients based on one’s unique needs.
Research continues to evolve, leading to deeper insights about gene-diet interactions. The potential for improved health outcomes through personalization makes this an exciting arena worth watching closely.
Why Considering Genetics in your Diet can Lead to Better Health Outcomes
Understanding your genetic makeup can transform how you approach nutrition. Each individual has unique genetic variations that influence metabolism, nutrient absorption, and even food preferences.
When you consider these factors in your diet, you gain a roadmap to better health. Tailoring meals to match your genetics can optimize energy levels and enhance overall well-being.
For example, some people may metabolize fats more efficiently than carbohydrates. By aligning dietary choices with this knowledge, they can achieve greater weight management success.
Moreover, recognizing predispositions to certain conditions empowers proactive measures. A personalized diet could reduce the risk of heart disease or diabetes by focusing on foods that support one's specific needs.
Incorporating genetics into nutritional planning fosters a deeper connection with food. It encourages mindful eating habits that resonate on an individual level while promoting healthier lifestyle choices over time.
Conclusion
Personalized nutrition is reshaping how we understand diet and health. By considering genetics, individuals can create tailored diets that better suit their unique needs. The insights gained from nutrigenomics are just the beginning, revealing a complex interplay between our genes and how we process various nutrients.
As more people turn to genetic testing for dietary guidance, success stories continue to emerge. These narratives highlight not only weight loss but also improved energy levels and overall well-being. Yet challenges remain in this evolving field—such as misinformation about genetic tests or the potential for over-reliance on technology.
Looking forward, advancements in personalized nutrition promise even greater precision in tailoring diet plans. As research progresses, it offers hope for healthier lifestyles through scientifically-backed approaches.
Embracing genetics as part of your nutritional strategy may lead to enhanced health outcomes and a deeper understanding of what works best for you personally. This exciting frontier invites everyone to explore their individual pathways toward optimal health through informed dietary choices.
Revolutionizing Healthcare: Top Medical Innovations
Revolutionizing Healthcare: Top Medical Innovations
The healthcare landscape is undergoing a seismic shift. Innovations that once seemed like science fiction are now transforming the way we diagnose, treat, and manage diseases. From artificial intelligence to gene editing, these advancements promise to improve patient outcomes and streamline medical processes. As we stand on the brink of a new era in medicine, it’s essential to explore the cutting-edge technologies reshaping our world. This journey into medical innovations reveals not just their potential but also their challenges. Let's delve deeper into how these breakthroughs are revolutionizing healthcare as we know it.
Understanding Medical Innovations
Medical innovations encompass a broad spectrum of advancements that enhance patient care and streamline healthcare delivery. These developments can include new technologies, techniques, or drugs designed to improve health outcomes.
At the heart of medical innovation is a relentless pursuit of solutions to existing problems. Whether it's finding more effective treatments for chronic diseases or improving surgical procedures, each breakthrough adds value to healthcare systems worldwide.
Collaboration among scientists, engineers, doctors, and patients fuels this progress. Clinical trials often serve as testing grounds where novel ideas are rigorously evaluated before reaching the mainstream market.
Moreover, understanding these innovations requires awareness of various factors such as regulatory standards and ethical considerations. As we explore specific breakthroughs further along in this article, it becomes clear how integral these innovations are in addressing some of healthcare's most pressing challenges.
Artificial Intelligence in Healthcare
Artificial Intelligence (AI) is transforming healthcare in remarkable ways. By analyzing vast amounts of data, AI can identify patterns that elude human eyes. This capability enhances diagnostics and treatment plans.
Machine learning algorithms are making waves in radiology. They assist radiologists by detecting anomalies in imaging scans with impressive accuracy. This leads to quicker diagnoses and improved patient outcomes.
Chatbots powered by AI are revolutionizing patient interactions too. They provide instant responses to common inquiries, easing the burden on healthcare staff. These virtual assistants enhance efficiency while maintaining a personal touch.
Moreover, predictive analytics helps hospitals manage resources more effectively. By forecasting patient volumes, facilities can optimize staffing and reduce wait times.
As these technologies evolve, the potential for personalized medicine expands exponentially. Tailoring treatments based on individual genetic profiles has never been more attainable thanks to AI advancements.
Virtual Reality for Pain Management
Virtual reality (VR) is changing the landscape of pain management. By immersing patients in digital environments, VR distracts them from their discomfort.
Imagine a patient undergoing physical therapy while exploring a serene beach or soaring through the sky. These experiences can significantly reduce perceived pain levels during treatment sessions.
Studies show that VR not only helps with acute pain but also aids in chronic conditions. Patients find themselves more relaxed and engaged, leading to better outcomes.
Healthcare providers are now incorporating this technology into various treatments. It offers an innovative approach without reliance on medication alone.
As VR continues to evolve, its applications in mental health and rehabilitation grow even wider. The potential for personalized experiences makes it an exciting frontier in medical innovation.
3D Printing in Medicine
3D printing is reshaping the medical landscape in astonishing ways. It allows for the creation of customized implants, prosthetics, and even organ models tailored specifically to each patient’s anatomy.
Surgeons can now practice complex procedures on 3D-printed replicas before operating on actual patients. This hands-on approach enhances precision and boosts confidence during real surgeries.
Additionally, bioprinting is emerging as a groundbreaking innovation. Researchers are exploring ways to print tissues and organs using living cells. The potential for transplantable organs made from a patient’s own cells could transform organ donation forever.
The cost-effectiveness of 3D printing also cannot be ignored. It streamlines production processes, reducing waste while delivering rapid results.
As technology improves, we may see more advanced applications in personalized medicine that were once only imagined in science fiction novels. The implications for enhanced patient care are boundless.
Gene Editing Technology
Gene editing technology, particularly CRISPR-Cas9, is transforming the landscape of medicine. This powerful tool allows scientists to modify DNA with incredible precision. By cutting out faulty genes and inserting healthy ones, it opens doors for treating genetic disorders.
The potential applications are staggering. Imagine curing inherited diseases like cystic fibrosis or sickle cell anemia at their source. Researchers are also exploring gene therapy for conditions that were once deemed untreatable.
Ethical implications arise as well. The ability to edit genes raises questions about designer babies and unforeseen consequences in ecosystems. These debates challenge us to consider the moral aspects alongside scientific advancements.
As this technology evolves, its accessibility will widen significantly. The future may see personalized treatments tailored specifically to an individual’s genetic makeup—a true revolution in healthcare delivery.
Non-invasive Diagnostics and Treatments
Non-invasive diagnostics and treatments are transforming how we approach patient care. Traditional methods often involve invasive procedures that can be daunting for patients. Now, advanced technologies offer alternatives that minimize discomfort.
Imaging techniques like MRI and ultrasound provide detailed insights without needing incisions. These tools allow physicians to visualize internal organs safely and effectively. Patients appreciate the reduced recovery times associated with such approaches.
Moreover, non-invasive treatments are gaining traction in areas like pain management and chronic disease therapy. Techniques such as focused ultrasound or transcutaneous electrical nerve stimulation (TENS) enable effective relief without surgery.
Wearable devices also play a vital role in this shift. They monitor health metrics continuously, giving real-time feedback to both patients and doctors. This data-driven approach enhances personalized care while reducing unnecessary interventions, paving the way for more holistic treatment plans tailored to individual needs.
Impact of Medical Innovations on Healthcare Industry
Medical innovations are reshaping the healthcare landscape in profound ways. They enhance patient care, streamline processes, and improve outcomes.
Take telemedicine as an example. It bridges geographical gaps, allowing patients to consult doctors without leaving their homes. This has made healthcare more accessible for many.
Wearable technology also plays a crucial role. Devices that monitor heart rates or track fitness levels empower individuals to take charge of their health proactively.
Moreover, data analytics enables providers to predict patient needs better and personalize treatment plans. The shift towards preventive care is transforming how we view health management.
As these innovations continue to evolve, they promise not only enhanced efficiency but also cost reductions across the board. Patients can expect quicker diagnoses and tailored therapies that cater specifically to them.
The ripple effect of these advancements means a healthier population overall, with fewer hospital visits and improved quality of life for countless individuals.
Ethical and Legal Concerns
As medical innovations surge forward, ethical and legal concerns loom large. The rapid advancement of technologies like AI and gene editing raises questions about consent and accountability.
Who is responsible when a machine makes an error? This uncertainty complicates the landscape for healthcare providers. They must navigate potential liabilities while trying to embrace cutting-edge solutions.
Privacy issues also come into play with non-invasive diagnostics. Patients' data is more accessible than ever, increasing the risk of breaches. Protecting sensitive information becomes paramount in this digital age.
Moreover, equity in access to these innovations remains a critical concern. Not all populations benefit equally from medical breakthroughs, leading to disparities that can exacerbate existing health inequalities.
Balancing innovation with ethical considerations will shape the future of healthcare as we know it. Careful thought and regulation are essential to ensure that progress does not come at too high a cost.
Future Possibilities and Advancements
The future of healthcare seems limitless. Innovations are on the horizon that promise to transform patient experiences and outcomes. Imagine a world where diseases can be diagnosed before symptoms even appear, thanks to advanced predictive analytics.
Wearable technology continues to evolve, providing real-time health monitoring that empowers individuals. These devices could soon integrate seamlessly with AI algorithms for personalized treatment plans tailored to each user's unique biology.
Telemedicine is also set for remarkable growth. As connectivity improves, remote consultations could become routine, breaking geographical barriers in medical access.
Additionally, breakthroughs in nanotechnology may enable targeted drug delivery systems. This advancement could minimize side effects while maximizing therapeutic effectiveness.
As these technologies converge, they will not only enhance efficiency but also reshape our understanding of wellness and disease management. The landscape is gearing up for an exciting transformation filled with possibilities yet to be fully realized.
Conclusion
The rapid pace of medical innovations is reshaping the healthcare landscape. From artificial intelligence that enhances diagnostics and treatment plans to virtual reality techniques used for pain management, these advancements are making a significant impact.
3D printing allows for customized prosthetics and even organ creation, while gene editing technologies like CRISPR hold promise for treating genetic disorders. Non-invasive diagnostic tools improve patient comfort and reduce recovery times.
While the benefits are clear, ethical considerations cannot be overlooked. Privacy concerns with AI algorithms and the potential misuse of gene editing technology raise questions about regulation and responsibility.
Looking ahead, the future holds immense possibilities. Continued investment in research can lead to breakthroughs we haven’t yet imagined. The integration of these innovations into everyday practice will ultimately enhance patient care but requires careful navigation through its complexities.
The journey of revolutionizing healthcare continues, paving new pathways toward a healthier society.
The Rise of Quantum Computing: Opportunities and Challenges
The Rise of Quantum Computing: Opportunities and Challenges
The world of computing is on the verge of a groundbreaking transformation. Enter quantum computing, a technology that promises to revolutionize how we process information. Imagine computers that can solve complex problems in seconds, ones that mimic the very laws of physics at an atomic level. As researchers and tech enthusiasts dive deeper into this innovative field, both opportunities and challenges emerge.
From enhancing artificial intelligence to cracking codes faster than ever before, quantum computing could reshape industries from finance to healthcare. But with great power comes great responsibility—alongside its potential lies a host of hurdles that must be navigated carefully. Join us as we explore this fascinating realm where science meets possibility and discover what it means for our future.
Understanding Quantum Computing
Quantum computing leverages the principles of quantum mechanics to process information in ways that classical computers can’t. At its core, it uses qubits—quantum bits—that exist in multiple states simultaneously thanks to superposition. This allows for an exponential increase in computational power.
Entanglement is another fundamental principle at play. When qubits become entangled, the state of one instantly influences the other, regardless of distance. This interconnectedness enables complex problem-solving capabilities far beyond traditional binary systems.
Unlike standard bits that represent a 0 or a 1, qubits can embody both values at once. This unique feature opens new avenues for calculations and simulations previously thought impossible.
As researchers unravel these complexities, they edge closer to unlocking the true potential of this technology—a leap into uncharted territories of computation and innovation.
The Advantages of Quantum Computing
Quantum computing offers unparalleled speed. Traditional computers process data in bits, while quantum computers use qubits. This allows them to handle complex calculations at lightning speed.
Another advantage lies in their ability to solve problems deemed impossible for classical systems. Quantum algorithms can tackle optimization issues and simulate molecular interactions more efficiently than ever before.
Moreover, quantum computing enhances security through advanced cryptography. With quantum encryption methods, information becomes virtually unhackable, providing a significant edge for sensitive data management.
The power of superposition enables multiple calculations simultaneously. This unique property greatly accelerates tasks such as drug discovery and financial modeling.
Furthermore, the potential for creating smarter AI is immense. Quantum machines could revolutionize machine learning by processing vast amounts of data more effectively than current technologies permit.
This technology promises not just advancements but entirely new paradigms across industries.
Real-World Applications of Quantum Computing
Quantum computing is making waves across various sectors. Its ability to process vast amounts of data at unprecedented speeds opens doors that classical computers can’t.
In finance, quantum algorithms are revolutionizing portfolio optimization. They analyze market trends and risks swiftly, giving firms a competitive edge.
Healthcare also benefits significantly. With quantum computing, researchers can simulate molecular interactions more accurately. This accelerates drug discovery and personalized medicine development.
Logistics companies leverage this technology for complex routing problems. By optimizing supply chains in real-time, they reduce costs and improve delivery times.
Even cybersecurity sees advancements through quantum encryption methods. These ensure secure communication by utilizing the principles of superposition and entanglement.
As industries explore these applications, the potential impact becomes clearer. The future seems bright for innovations driven by quantum technologies across multiple fields.
Challenges Facing Quantum Computing
Quantum computing is an exciting frontier, but it comes with notable challenges. One significant hurdle is qubit stability. Qubits are highly sensitive to their environment, leading to errors during calculations.
Another issue lies in the complexity of quantum algorithms. Developing these algorithms requires specialized knowledge that’s still evolving within the tech community.
Scalability also poses a major challenge. Current quantum systems can handle only a limited number of qubits efficiently. Expanding this capability without compromising performance is crucial for practical applications.
Additionally, there’s the question of integration with existing technologies. Businesses need solutions that can seamlessly work alongside classical computing systems.
Security concerns cannot be overlooked. Quantum computers have the potential to break traditional encryption methods, prompting a race for new cryptographic techniques that can withstand such advancements.
The Future of Quantum Computing
Quantum computing is on the brink of revolutionizing how we approach complex problems. As research accelerates, breakthroughs are becoming more frequent. This rapid progression hints at a future where quantum computers become mainstream.
Imagine solving intricate equations in seconds that currently take classical machines years. Industries like finance and healthcare could transform their operations dramatically.
The potential for advancements in artificial intelligence is equally compelling. Quantum systems can process vast datasets with unmatched speed, leading to smarter algorithms and predictive models.
Moreover, as companies invest heavily in developing quantum infrastructure, collaboration will grow across sectors. Startups and established firms will unite to harness this technology's capabilities.
Regulatory frameworks will evolve too, ensuring ethics keep pace with innovation. The landscape of data security might change entirely as quantum encryption offers unprecedented protection against cyber threats.
This evolution opens new doors for businesses ready to adapt and innovate within this dynamic environment.
Opportunities for Businesses and Industries
Quantum computing opens a new frontier for businesses across various sectors. Industries like finance can harness its power to optimize portfolios and manage risk more effectively.
Healthcare also stands to benefit significantly. Quantum algorithms might revolutionize drug discovery, enabling faster development of life-saving medications.
Manufacturing could see improvements in supply chain management. By processing vast amounts of data swiftly, companies can forecast demand with greater accuracy.
Telecommunications may leverage quantum encryption technologies, enhancing security protocols against cyber threats. This advancement is crucial in an era of increasing digital vulnerabilities.
Moreover, the entertainment industry might explore enhanced graphics rendering or complex simulations through quantum capabilities, leading to richer user experiences.
As organizations begin adapting these innovations, collaboration between tech firms and established industries will create even more opportunities for growth and transformation.
Conclusion
Quantum computing is on the brink of revolutionizing how we process information. As researchers continue to unlock its potential, both opportunities and challenges emerge. The technology promises faster processing capabilities that could transform industries like healthcare, finance, and logistics.
However, significant hurdles remain. From technological limitations to ethical considerations surrounding data security and privacy, these challenges must be addressed for quantum computing to reach its full potential.
Businesses should begin exploring ways to integrate this innovative technology into their operations now. By investing in research or collaborating with tech companies specializing in quantum innovation, organizations can position themselves at the forefront of this new wave.
The journey of quantum computing has just begun. It's an exciting time for technologists and businesses alike as they navigate through uncharted waters filled with promise and possibility. Embracing change may lead to groundbreaking advancements that reshape our future.
Innovations in Food Packaging Technology
Innovations in Food Packaging Technology
Food packaging is often overlooked, yet it plays a crucial role in our daily lives. It protects our meals, preserves freshness, and provides essential information. But what if food packaging could do more? With rapid advancements in technology, the industry is evolving at an unprecedented pace. Innovations are reshaping how we think about packaging—making it smarter, more sustainable, and even interactive.
Imagine grabbing a snack that not only tastes great but also informs you of its nutritional benefits or alerts you when it's nearing expiration. As consumers become increasingly environmentally conscious and tech-savvy, the demand for innovative solutions grows stronger than ever.
Let's dive into the exciting world of food packaging technology and explore how these innovations are revolutionizing what we eat and how we experience food!
Understanding Food Packaging Technology
Food packaging technology encompasses various methods and materials designed to protect food from contamination, spoilage, and damage during storage and transport. It goes beyond aesthetics; it’s about functionality.
At its core, effective packaging preserves the freshness of products while ensuring safety. This is achieved using barriers to oxygen, moisture, and light. Each factor plays a significant role in extending shelf life.
Moreover, advancements have led to innovations like vacuum sealing and modified atmosphere packaging (MAP). These techniques adjust the composition of gases surrounding food items to slow down deterioration processes.
The integration of smart technologies—like RFID tags—is also gaining traction. These enable real-time tracking of inventory and temperature control throughout the supply chain.
With consumers demanding transparency regarding their food sources, understanding the science behind food packaging is vital for both manufacturers and buyers alike.
Benefits of Innovative Food Packaging
Innovative food packaging brings a multitude of advantages to both producers and consumers. Enhanced shelf life is one of the most significant benefits. Advanced materials help keep products fresher for longer, reducing waste.
Another key aspect is safety. Improved sealing techniques and barrier properties protect against contaminants, ensuring that food remains safe and free from spoilage. This enhances consumer confidence in the products they choose.
Additionally, eye-catching designs and features can elevate brand identity. Packaging that stands out on shelves often leads to increased sales as it attracts more attention from potential buyers.
Cost efficiency also plays a role in innovative packaging solutions. By using lighter materials or optimizing sizes, companies can reduce shipping costs while minimizing their environmental footprint.
With technological advancements evolving rapidly, these benefits will only expand further in scope, making food packaging an exciting field to watch closely.
Sustainable Packaging Solutions
Sustainable packaging solutions are transforming the food industry. Companies are now prioritizing materials that minimize environmental impact.
From recycled paper to plant-based plastics, choices abound. These options not only reduce waste but also lower carbon footprints.
Innovative designs play a crucial role too. Brands are reconsidering shapes and sizes to use less material while maintaining product integrity.
Consumer demand for eco-friendly practices is rising, pushing businesses to adapt quickly. Many brands highlight their sustainability efforts on packaging, appealing directly to environmentally conscious shoppers.
These initiatives also foster brand loyalty. Customers appreciate transparency and responsibility in their purchasing decisions.
As regulations tighten around single-use plastics, companies must innovate or risk falling behind. Sustainable solutions aren't just a trend; they’re becoming essential for long-term success in the market.
Smart and Interactive Packaging Options
Smart packaging is transforming how consumers interact with products. Imagine a cereal box that can tell you when it’s running low or a milk carton that shows its freshness status. These innovations utilize sensors and QR codes to provide real-time information.
Interactive packaging engages customers in new ways. Scanning a code may lead to recipes, nutritional information, or even promotional offers. This connectivity adds value beyond the product itself.
Moreover, smart options help brands gather data on consumer behavior. Understanding preferences allows companies to tailor marketing strategies effectively.
Sustainability also plays a role here. Some smart packages are designed to be reused or recycled easily, minimizing waste while enhancing user experience.
As technology advances, expect more creative solutions that keep customers informed and engaged right at their fingertips.
Biodegradable and Compostable Packaging Materials
The shift towards biodegradable and compostable packaging materials is gaining momentum. These alternatives break down naturally, reducing landfill waste and environmental impact.
Biodegradable materials decompose through natural processes, often within months. This means less plastic lingering in our ecosystems. Compostable options take it a step further, turning into nutrient-rich soil when processed correctly.
Made from plant-based resources like cornstarch or sugarcane, these products are not only eco-friendly but also versatile. Brands are increasingly adopting them to meet consumer demand for sustainable practices.
However, proper disposal is crucial for their effectiveness. Many consumers still struggle with the right recycling methods. Education on how to handle these materials can enhance their benefits significantly.
As technology advances, expect even more innovative solutions in this realm. The future of food packaging could be greener than ever before.
Nanotechnology in Food Packaging
Nanotechnology is revolutionizing food packaging in remarkable ways. By manipulating materials at the molecular level, scientists create packaging that enhances food safety and shelf life.
These innovations often involve nanomaterials that can improve barrier properties, making it tougher against moisture and oxygen. This means less spoilage and longer-lasting freshness for consumers.
Additionally, some nano-packaging incorporates antimicrobial agents. These tiny particles effectively inhibit the growth of bacteria on food surfaces, ensuring safer consumption without compromising taste or quality.
Another exciting aspect is the development of smart packaging with embedded nanosensors. These sensors monitor conditions like temperature and humidity, providing real-time feedback about the product's state.
As we embrace advanced technologies in our daily lives, nanotechnology stands out as a key player in elevating food packaging solutions to new heights. It opens doors to safer, more efficient options for both producers and consumers alike.
Future Trends in Food Packaging Technology
The future of food packaging technology is poised for exciting advancements. One notable trend is the integration of artificial intelligence to enhance supply chain management. AI can predict demand, reducing waste and optimizing production.
Another emerging development focuses on minimalist packaging designs. These aim to reduce material usage while maintaining product integrity. This not only appeals to eco-conscious consumers but also addresses cost efficiency.
Moreover, 3D printing technology holds great promise for creating customized packaging solutions tailored to specific products or brands. This could revolutionize how businesses approach design and manufacturing processes.
Additionally, augmented reality (AR) is making its way into food packaging. Consumers can scan packages with their smartphones to access nutritional information, recipes, or even interactive experiences related to the brand.
As sustainability remains a priority, innovations in edible packaging may become mainstream. Imagine enjoying your snack without any waste at all—just eat the wrapper too!
Conclusion
The landscape of food packaging technology is evolving rapidly. Innovations are not just enhancing functionality; they’re also transforming how consumers interact with products. As sustainability becomes a priority, brands are rethinking their materials and processes to reduce environmental impact. The rise of smart packaging offers exciting possibilities, allowing for better tracking and enhanced customer engagement.
As we look ahead, the integration of nanotechnology promises even more breakthroughs in safety and preservation. With each advancement, we're moving closer to a future where food remains fresher longer while being kinder to our planet.
Navigating this new world requires awareness and adaptation from both consumers and producers alike. Keeping up with these trends ensures that everyone can play a part in fostering sustainable practices while enjoying the benefits of innovative packaging solutions. The journey of food packaging continues to inspire creativity and responsibility within the industry—shaping how we think about what we eat and how it reaches us.
Innovative Materials Shaping the Future
Innovative Materials Shaping the Future
The world around us is constantly evolving, and at the heart of this transformation lies a fascinating realm of innovative materials. From the clothes we wear to the buildings we inhabit, these materials are reshaping our lives in ways we never thought possible. Imagine living in homes made from carbon-capturing bricks or using packaging that disappears without a trace. The future is not just a distant dream; it's happening right now.
As technology advances and sustainability becomes paramount, new materials are emerging to meet our needs while protecting our planet. This blog post will explore some of these groundbreaking innovations and their potential impact on various industries—from construction to healthcare. Join us as we dive into the exciting world of materials that promise to redefine how we live, work, and interact with our environment!
Understanding Innovative Materials
Innovative materials are the building blocks of progress. They represent a shift in how we think about functionality, sustainability, and design.
These materials often combine cutting-edge science with creative engineering. They can be lightweight yet incredibly strong or flexible while maintaining structural integrity. This versatility opens up new possibilities across various fields.
Consider graphene, for example—a single layer of carbon atoms that boasts remarkable strength and conductivity. Its potential applications span from electronics to renewable energy technologies.
Equally exciting are bio-inspired materials that mimic nature’s designs for improved efficiency and durability. By studying organisms like spider silk or lotus leaves, researchers create solutions that enhance our everyday lives.
Understanding these innovations is crucial as they pave the way for smarter products and more sustainable practices. Each breakthrough offers a glimpse into what tomorrow might hold—an endless horizon of creativity waiting to be explored.
Advancements in Sustainable Materials
Sustainable materials are transforming industries. Innovations in this field focus on reducing environmental impact while maintaining functionality.
Bioplastics, derived from renewable resources, have gained traction. They replace traditional petroleum-based plastics and can be composted after use. This shift not only conserves fossil fuels but also addresses plastic waste issues.
Another exciting development is the rise of recycled materials. Companies are now sourcing post-consumer products to create new items, minimizing landfill contributions and promoting a circular economy.
Natural fibers like hemp and bamboo are also emerging as sustainable alternatives in textiles. These materials require less water and chemicals compared to conventional fabrics, making them kinder to our planet.
Moreover, advancements in bio-composites enhance strength while being lightweight. They offer an eco-friendly solution for various applications without compromising quality or efficiency.
These innovations highlight how sustainability is becoming integral to material science, paving the way for greener solutions across multiple sectors.
Impact of Biodegradable Materials on the Environment
Biodegradable materials are revolutionizing how we approach waste management. Unlike traditional plastics, these materials break down naturally over time, reducing landfill accumulation.
When exposed to environmental elements like moisture and microorganisms, they decompose into non-toxic substances. This process minimizes pollution and limits harmful effects on wildlife.
Moreover, the use of biodegradable options encourages responsible consumption. Consumers are becoming more aware of their choices and opting for products that align with eco-friendly values.
Industries are responding to this shift by innovating sustainable packaging solutions. From compostable bags to plant-based containers, businesses recognize the importance of protecting our planet.
While challenges remain in terms of production scalability and consumer education, the potential benefits make it a promising avenue worth exploring further. The movement towards biodegradable alternatives is not just a trend; it's an essential step toward sustainability.
Technological Breakthroughs in Smart Materials
Smart materials are revolutionizing industries with their remarkable adaptability and functionality. These materials respond dynamically to changes in their environment, allowing for more efficient designs.
One exciting advancement is shape memory alloys. They can return to a predetermined shape when heated, making them ideal for applications ranging from medical devices to aerospace engineering.
Another breakthrough involves piezoelectric materials that generate electric charge under mechanical stress. This property enhances energy harvesting technologies and opens doors for self-powered devices.
Moreover, advancements in thermochromic materials allow surfaces to change color based on temperature fluctuations. From building facades that adjust heat absorption to fashion that responds to body temperature, the potential applications are endless.
These innovations not only bridge gaps between technology and material science but also pave the way for smarter, more responsive products that enhance our daily lives. The possibilities continue expanding as research progresses at an astonishing pace.
The Use of Nanotechnology in Building and Manufacturing
Nanotechnology is revolutionizing building and manufacturing processes. By manipulating materials at the atomic or molecular level, it opens doors to unprecedented possibilities.
In construction, nanomaterials enhance strength while reducing weight. Imagine concrete that’s more durable yet lighter. This innovation leads to safer structures with lower environmental impact.
Manufacturers also benefit greatly from nanotechnology. It allows for precise control over material properties, leading to products that last longer and perform better. Coatings infused with nanoparticles can resist scratches or repel dirt effectively.
This technology doesn’t stop there; it's paving the way for energy-efficient systems as well. Nanotech-enabled insulation keeps buildings warmer in winters and cooler during summers without hefty energy bills.
The integration of nanotechnology into everyday applications is not just a trend—it's a glimpse into a future where efficiency meets sustainability effortlessly.
Innovative Materials for Medical Applications
Innovative materials are revolutionizing the medical field. From advanced polymers to bioactive glass, these substances are enhancing patient care.
3D-printed implants made from titanium alloys offer custom solutions for individual patients. They integrate seamlessly with bone tissue, promoting faster healing and reducing complications.
Additionally, hydrogels are gaining traction in drug delivery systems. Their ability to mimic natural tissues allows for targeted therapy and minimizes side effects.
Smart textiles infused with sensors monitor vital signs in real-time. This innovation promises proactive health management, leading to quicker interventions when needed.
Moreover, biodegradable sutures eliminate the need for removal procedures. They dissolve naturally within the body while supporting tissue repair.
These advancements highlight a shift towards personalized medicine and improved outcomes across various treatments. As research continues, the potential applications seem limitless.
Challenges and Concerns with Adopting New Materials
Adopting new materials comes with its own set of challenges. One significant hurdle is the cost associated with research and development. Innovative solutions often require hefty investments, which can be daunting for many businesses.
Additionally, there’s a learning curve involved in integrating these materials into existing production processes. Training staff to handle novel substances or technologies can disrupt operations and lead to temporary inefficiencies.
Regulatory hurdles also pose a challenge. New materials must meet various safety and environmental standards before they can hit the market. This lengthy approval process can delay project timelines significantly.
Moreover, there's resistance from traditional industries accustomed to established practices. Convincing stakeholders of the benefits offered by innovative alternatives requires strong evidence and persistent advocacy.
Public perception plays a role in adoption as well. Consumers are sometimes skeptical about new products, fearing unknown risks or potential harm to their health or environment.
Embracing the Future of Materials
The future of materials is bright and filled with potential. As industries evolve, so do the innovations that drive them forward.
Embracing new materials means challenging traditional practices. It opens doors to creativity and sustainability. Designers and engineers are eager to explore what these advancements can offer.
Collaboration across fields fosters breakthroughs. Scientists, manufacturers, and environmentalists are working together like never before. This synergy fuels developments that were once thought impossible.
Consumers also play a vital role in this shift. Increased awareness about sustainable options encourages demand for innovative materials. The market responds by prioritizing eco-friendly solutions.
As we adopt these novel substances, we redefine our relationship with technology and nature alike. Every step toward embracing change brings us closer to a more resilient world where innovation knows no bounds.
Conclusion
Innovative materials are not just a trend; they represent the evolution of industries and lifestyles. As we explore sustainable options, biodegradable alternatives, and smart technologies, it becomes clear that these advancements hold the potential to reshape how we interact with our environment.
The integration of nanotechnology is paving new pathways in construction and manufacturing. This allows for more efficient processes while enhancing performance. Meanwhile, medical applications demonstrate remarkable progress, improving patient care and treatment outcomes.
Yet challenges persist. Concerns regarding scalability, cost-effectiveness, and environmental impacts need addressing as we embrace these new materials. The road ahead will require collaboration between researchers, manufacturers, and policymakers to ensure responsible adoption.
As society continues to innovate at a rapid pace, keeping an eye on material science will be crucial for building a sustainable future. With each breakthrough comes the promise of improved quality of life—one innovative material at a time.
The Ultimate Guide to Using Epsom Salt in Your Garden
The Ultimate Guide to Using Epsom Salt in Your Garden
If you’re a garden enthusiast or just someone who loves to nurture plants, you’ve probably heard whispers about the magic of Epsom salt. But what exactly is this household staple doing in your garden? It’s more than just a bath-time treat; it’s packed with benefits that can enhance plant growth and health! Imagine lush greens, vibrant blooms, and bountiful harvests—all thanks to a simple magnesium sulfate compound.
This guide will delve into how Epsom salt works wonders for your plants, practical ways to incorporate it into your gardening routine, and even some common pitfalls to avoid. Whether you're growing vegetables or flowers, understanding the role of Epsom salt could be the secret ingredient that takes your garden from ordinary to extraordinary. Let’s dig deeper!
What is Epsom Salt and How Does it Benefit Plants?
Epsom salt, chemically known as magnesium sulfate, is a naturally occurring mineral compound. It's not just for soothing sore muscles; it can be a game-changer for your garden too.
Plants thrive on essential nutrients, and magnesium is one of them. Epsom salt provides this nutrient in an easily absorbable form. When plants have adequate magnesium levels, they can produce chlorophyll more efficiently. This means greener leaves and healthier growth overall.
Additionally, Epsom salt aids in the absorption of other key elements like nitrogen and phosphorus. It enhances seed germination and boosts flower production by promoting stronger root development.
Using Epsom salt isn't just about adding minerals; it's about creating an environment where plants can flourish naturally. Whether you're dealing with stubborn soil or seeking vibrant blooms, incorporating this simple ingredient could make all the difference in your gardening success.
The Role of Magnesium in Plant Growth
Magnesium is a vital nutrient for plants, playing several essential roles in their growth and development. It serves as the core component of chlorophyll, the pigment responsible for photosynthesis. Without magnesium, plants cannot effectively convert sunlight into energy.
This mineral also aids in the synthesis of enzymes that facilitate important biochemical reactions within plants. These processes include carbohydrate metabolism and protein synthesis.
Moreover, magnesium helps improve nutrient uptake from the soil. It assists in activating specific enzymes that enhance root growth and overall plant vigor.
A deficiency can lead to yellowing leaves and poor fruit production. Ensuring adequate magnesium levels through Epsom salt can significantly boost plant health and yields. This simple addition may transform your garden into a thriving oasis filled with vibrant foliage and bountiful harvests.
How to Use Epsom Salt in Your Garden
Epsom salt is a versatile tool for gardeners looking to enhance plant health. One of the simplest ways to use it is as a soil amendment. Just mix about one tablespoon per foot of plant height into the soil before planting. This gives your plants an immediate boost.
For those hard-to-reach leaves, consider making a foliar spray. Combine two tablespoons of Epsom salt with a gallon of water and spray directly onto foliage once a month. This method allows plants to absorb nutrients quickly.
If you're starting seeds, soak them in an Epsom salt solution beforehand. Mix two teaspoons in a quart of water and let the seeds soak for 24 hours. This encourages strong early growth by giving seedlings essential nutrients right from the start. Each method offers unique benefits tailored to different gardening needs, allowing you to choose what works best for your garden's specific conditions.
A. Soil amendment
Epsom salt can work wonders as a soil amendment. It’s rich in magnesium and sulfate, two essential nutrients that promote healthy plant growth.
When mixed into the soil, Epsom salt helps improve nutrient absorption. This allows plants to take in other vital minerals more effectively.
To use it as a soil amendment, simply mix about one tablespoon of Epsom salt per foot of plant height into the top few inches of your garden bed. You can also dissolve it in water for an easier application.
Applying Epsom salt before planting gives seeds and young plants a solid nutritional foundation. This small change can lead to stronger roots and vibrant foliage throughout the growing season.
Keep an eye on your plants after application; you may notice improved leaf color and overall vitality!
B. Foliar spray
Foliar spraying is an effective way to deliver nutrients directly to your plants. By applying Epsom salt as a foliar spray, you ensure that magnesium and sulfate are quickly absorbed through the leaves.
To make a simple solution, dissolve two tablespoons of Epsom salt in a gallon of water. Stir well until completely dissolved.
Using a spray bottle or garden sprayer, mist the mixture onto the foliage during cooler parts of the day—early morning or late afternoon works best. This timing helps prevent leaf burn while maximizing absorption.
Focus on the undersides of leaves where stomata are located for optimal nutrient uptake. Regular applications every few weeks can boost plant health and vigor.
Watch closely for changes in your plants' growth patterns after treatment; healthy green leaves and robust blooms often indicate success!
C. Seed soak
Soaking seeds in Epsom salt can give them a vital head start. This method is simple yet effective, enhancing both germination and growth.
To prepare your seed soak, mix about one tablespoon of Epsom salt with a quart of water. Stir until the salt dissolves completely. Soak your seeds in this solution for 24 hours before planting.
This process helps the seeds absorb magnesium efficiently. As they sprout, they'll benefit from improved nutrient uptake. You'll often notice stronger plants that are more resilient to stressors like drought or pests.
Just remember not to overdo it; excessive use may lead to imbalances in your soil's nutrients. A little goes a long way when working with nature’s delicate balance!
Common Mistakes When Using Epsom Salt in the Garden
Many gardeners make the mistake of overusing Epsom salt, thinking more is better. Excess magnesium can lead to nutrient imbalances in the soil, affecting plant health.
Another common error is applying it at the wrong time. Timing matters when using Epsom salt. It’s best applied during key growth stages rather than randomly throughout the season.
Some enthusiasts overlook soil testing. Without knowing your soil's current nutrient levels, you might be adding what your plants don’t need.
Mixing Epsom salt with regular fertilizers can also create issues. This combination isn’t always beneficial and may result in uneven nutrient distribution.
Not paying attention to plant types could hinder results. Different plants have varying magnesium needs; understanding these differences will enhance effectiveness in your garden.
DIY Epsom Salt Fertilizer Recipes
Creating your own Epsom salt fertilizer is a simple and rewarding process. Start with the basic mix: combine one tablespoon of Epsom salt with one gallon of water. This solution can be used as a foliar spray or soil drench.
For plants that crave nitrogen, blend two tablespoons of Epsom salt with one cup of coffee grounds and three cups of water. Let this steep for 24 hours before applying it to your garden beds.
Another effective recipe involves mixing equal parts Epsom salt, baking soda, and sugar. Dissolve these in about two gallons of warm water to create a nutrient-rich concoction perfect for tomatoes and peppers.
Experimenting with these recipes not only boosts plant health but also enriches the gardening experience. Customizing your fertilizer allows you to cater specifically to the needs of your plants while making use of readily available ingredients.
Tips for Choosing and Storing Epsom Salt
When selecting Epsom salt, opt for a high-quality grade. Look for options labeled as “pure” or “food grade.” These varieties are free from additives and impurities that can harm your plants.
Consider the packaging too. Choose resealable bags or containers to keep moisture out. Humidity can clump Epsom salt, making it harder to use when you need it.
Store your Epsom salt in a cool, dry place away from direct sunlight. This helps maintain its effectiveness and prolongs shelf life.
If you’re using larger quantities, bulk purchases might be cost-effective. Just ensure that whatever size you choose is easy to handle in your garden routine.
Always label your storage container clearly if you're mixing with other gardening supplies. Clear identification prevents confusion later on during application days!
Conclusion
Epsom salt is a versatile and beneficial addition to any garden. Its high magnesium content plays a vital role in plant health, supporting growth and enhancing nutrient absorption. Whether you're amending the soil, applying it as a foliar spray, or soaking seeds before planting, Epsom salt can significantly impact your plants' performance.
While using Epsom salt offers many advantages, it's important to avoid common mistakes that could hinder its effectiveness. Be mindful of how much you apply and consider the specific needs of different plants.
Creating DIY Epsom salt fertilizer recipes allows gardeners to tailor their approach based on individual requirements. Plus, knowing how to choose and store Epsom salt properly ensures that it remains effective when needed.
Incorporating this simple ingredient into your gardening routine can lead to healthier plants and bountiful harvests. With proper use and awareness of best practices, you'll see just how transformative Epsom salt can be for your garden's success.
Smart Cities: Integrating Technology and Urban Planning
Smart Cities: Integrating Technology and Urban Planning
Picture this: a city where traffic flows smoothly, waste is managed efficiently, and energy consumption is optimized. This isn’t the plot of a futuristic movie; it’s the reality of smart cities. These urban environments harness cutting-edge technology to enhance the quality of life for their residents while promoting sustainability.
Smart cities combine data analytics, Internet of Things (IoT) devices, and innovative infrastructure to create interconnected communities that thrive on efficiency and convenience. As urban populations continue to swell, the need for effective solutions becomes urgent. Enter smart cities—transformative spaces redefining how we live, work, and interact in an increasingly digital world.
Curious about what makes these cities tick? Let’s explore the benefits they offer and examine successful initiatives around the globe that are paving the way for smarter living.
What are Smart Cities?
Smart cities epitomize the fusion of technology and urban life. They leverage data and connectivity to improve the daily experiences of residents while addressing pressing challenges.
At their core, smart cities utilize a network of sensors, devices, and applications. These tools gather real-time information about everything from traffic patterns to energy usage. This data-driven approach allows for informed decision-making.
Imagine a city where public transportation is optimized based on commuter habits or waste bins signal when they need emptying. Such advancements lead to enhanced efficiency in resource management.
Moreover, smart cities prioritize citizen engagement through accessible platforms that encourage participation in local governance. This creates a more responsive environment tailored to community needs.
These innovative spaces aim not just for technological advancement but also for social equity and environmental sustainability. The vision is clear: smarter solutions for better urban living.
Benefits of Smart Cities
Smart cities bring a multitude of benefits that enhance urban living. By leveraging technology, they improve the efficiency of services like waste management and public transportation.
With real-time data analysis, traffic congestion can be reduced significantly. This leads to shorter commute times and less environmental impact from vehicles.
Public safety is another advantage. Smart surveillance systems help monitor crime hotspots, allowing for quicker police response times.
Additionally, smart energy grids optimize electricity usage, reducing costs for residents while promoting sustainability. Efficient water management systems conserve resources and ensure clean drinking water.
The integration of green spaces in these cities improves air quality and boosts community well-being. All age groups find more opportunities for recreation and social interaction through enhanced city design.
Smart cities foster economic growth by attracting businesses eager to invest in innovative infrastructures.
Examples of Successful Smart City Projects
Barcelona stands out as a pioneer in smart city initiatives. The city has implemented an extensive network of sensors that monitor air quality, traffic conditions, and even waste management. This data helps optimize resources and improve the overall urban experience.
Singapore is another shining example. Its Smart Nation program integrates cutting-edge technology into daily life. From cashless payments to autonomous vehicles, Singapore leverages innovation to enhance mobility and efficiency.
In Amsterdam, sustainability takes center stage. The city encourages bike-sharing programs and smart energy solutions, creating a greener environment while promoting healthy lifestyles.
Songdo in South Korea was built from scratch with smart features embedded throughout its design. Every aspect—from trash collection to transportation—is managed through advanced technologies aimed at reducing environmental impact and improving livability for residents.
These projects demonstrate the potential of smart cities to transform urban living through thoughtful integration of technology.
Challenges and Criticisms of Smart Cities
Smart cities face significant challenges that can hinder their development. One major concern is data privacy. As cities collect vast amounts of information from citizens, questions arise about who owns this data and how it’s used.
Another issue is the digital divide. Not all residents have equal access to technology or the internet. This disparity can exacerbate existing social inequalities, leaving some communities behind in the smart city evolution.
Moreover, there are concerns about over-reliance on technology. Systems can fail or be vulnerable to cyberattacks, leading to disruptions in essential services such as transportation and healthcare.
Public engagement often falls short. Residents may feel disconnected from decision-making processes regarding smart initiatives, which could result in a lack of trust between citizens and local governments. Addressing these criticisms is crucial for creating truly effective smart cities that serve everyone equitably.
The Role of Technology in Urban Planning
Technology is revolutionizing urban planning in unprecedented ways. It provides tools that help city planners make informed decisions for sustainable development.
Geographic Information Systems (GIS) allow for detailed mapping and analysis of urban areas. Planners can visualize data to identify trends, such as population growth or traffic patterns. This insight helps create efficient infrastructure solutions.
Smart sensors are another game changer. They collect real-time data on air quality, noise levels, and energy consumption. With this information, cities can respond quickly to issues like pollution or congestion.
Drones offer a bird's-eye view of urban landscapes. They assist in surveying land and monitoring construction projects from above, ensuring compliance with regulations.
Moreover, technology fosters community engagement through apps and online platforms. Citizens can share feedback on projects or report issues directly to local authorities, enhancing transparency and collaboration in the planning process.
Collaboration between Government and Tech Companies
Collaboration between government and tech companies marks a pivotal shift in urban development. These partnerships foster innovative solutions tailored to the unique challenges cities face today.
Tech firms bring expertise in data analytics, artificial intelligence, and smart infrastructure. Governments provide regulatory frameworks, funding, and community insights. Together, they create smarter public services that enhance daily life.
Public-private partnerships can streamline processes. They enable rapid deployment of technology-driven projects like traffic management systems or energy-efficient buildings.
Open dialogue is crucial for success. Both parties must address concerns regarding privacy and security while prioritizing citizens' needs. Transparency builds trust among residents who rely on these initiatives.
Effective collaboration paves the way for sustainable growth and improved quality of life within urban environments. Engaging local communities ensures that technological advancements resonate with their specific needs.
Building a Sustainable and Inclusive Smart City
Building a sustainable and inclusive smart city requires careful planning. It involves integrating green technologies while ensuring accessibility for all residents.
Urban spaces should prioritize renewable energy sources, like solar panels and wind turbines. These solutions not only reduce carbon footprints but also create jobs in the green sector.
Inclusivity is equally essential. Smart cities must cater to diverse populations by considering various needs—ranging from affordable housing to accessible public transit systems. Input from community members can guide decision-making processes.
Public spaces equipped with Wi-Fi and tech resources help bridge the digital divide, enabling everyone to thrive in this modern environment.
Investing in education about technology fosters equal opportunities for citizens of all backgrounds. This empowers individuals to engage fully in their communities and local economies.
A successful smart city balances innovation with social responsibility, creating an urban landscape where everyone can flourish together.
The Future of Smart Cities
The future of smart cities is bright and full of potential. As technology evolves, urban landscapes will transform into interconnected ecosystems.
Imagine streets equipped with sensors that optimize traffic flow in real-time. This could reduce congestion and lower emissions dramatically, creating cleaner air for residents.
Data-driven decision-making will become the norm. City planners will harness analytics to enhance public services like waste management and energy use.
Moreover, citizen engagement will be at the forefront. Residents will have platforms to voice concerns or suggest improvements directly linked to city officials.
Inclusivity is another critical aspect. Smart cities must ensure that advancements benefit all citizens, regardless of their socioeconomic background.
As we embrace innovation, sustainability must remain a priority—balancing progress with environmental stewardship is essential for long-term success in urban development.
Conclusion
Smart cities represent a transformative vision for urban living. They harness technology to create more efficient, sustainable, and inclusive environments. As cities around the globe embrace this concept, they are not just solving existing problems but also paving the way for future innovations.
The benefits of smart cities are compelling. From reducing traffic congestion through intelligent transport systems to enhancing public safety with real-time surveillance technologies, these initiatives can significantly improve quality of life. Moreover, successful projects from places like Barcelona and Singapore showcase how integrated solutions can lead to measurable results.
However, challenges remain. Issues such as data privacy concerns and the digital divide must be addressed if we want all citizens to benefit equally from these advancements. The collaboration between government entities and tech companies is key in overcoming these hurdles while ensuring that technological integration aligns with urban planning goals.
Sustainability is another crucial aspect that cannot be overlooked when designing smart cities. By focusing on renewable resources and green spaces, we can achieve an environment where both nature and technology coexist harmoniously.
As we look ahead, it's clear that the evolution of smart cities will play a pivotal role in shaping our societies. With continued innovation and thoughtful planning, there’s potential not only for enhanced urban experiences but also for fostering communities where everyone thrives together.







