Fundamental Quantities and Units
Introduction
Measurements are part and parcel of everyday life. You may have weighed masses and volume of different substances e.g. 1kg of sugar, 500g of flour, 2litres of water. All substances that you are able to measure are called physical quantities.
In most cases measurements that we take are associated with errors. In actual experiments it is very difficult to record 100% accurate measurements. This is the case due errors that may be associated either with you as a researcher or the instruments being used. All measurements of a physical quantity that you take in science are recorded in SI units.
Physics is a branch of science that deals with measurements. It involves scientific procedures and processes that deal with error analysis in measurements and standard presentation of figures in science. Your understanding of this unit is very important to both Physics and General Science.
Objectives
By the end of this article you should be able to:
- Define physical quantities
- Differentiate derived quantities from basic quantities
- Analyze dimensions of different physical quantities
- Evaluate errors associated physical quantities
Physical Quantities
A physical quantity is a quantity that can be measured. In your homes, schools and in everyday life you take measurements of maize flour, sugar, water, paraffin, etc. All these are examples of physical quantities. We can classify physical quantities in two, scalar quantities and vector quantities.
Scalar Quantities
Quantities that have magnitude only, e.g. mass, temperature, time, length. The magnitude of a scalar quantity indicates its numerical value and unit of measure.
Vector Quantities
Quantities that have both magnitude and direction, e.g. velocity, force, momentum, displacement, etc. The magnitude of a vector is a scalar that indicates only how large or small the vector is.
Units
A quantity used as a standard of measurement is called a unit. In Physics we use two sets of units, thus basic units and derived units.
Basic Units
Basic units are units selected for measuring mass, length and time. These quantities cannot be expressed in terms of other quantities. We have seven basic quantities in physics. The units of all the other physical quantities in both science and engineering are derived from these seven basic quantities.
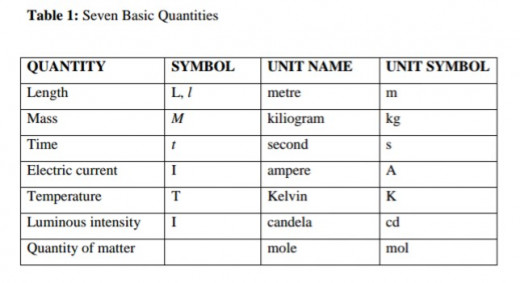
Basic Quantities
Derived Units
Having looked at basic or fundamental units now we want to consider a combination of several basic units. Units obtained from a combination of several basic units through multiplication and division are referred to as derived units. You may alternatively define derived units as units of physical quantities that can be expressed in terms of fundamental units. We normally use units of length, mass and time to come up with derived units.
We have plenty of examples of derived units that we encounter in everyday life for example, velocity, force, work, etc.
Example 1, Show that the unit of force is a derived quantity
Solution:
Velocity = displacement/time= length/time=m/s
Multiples and sub-multiples of SI Units
We can generate multiples and sub-multiples of SI units by adding appropriate prefixes to the units. Every prefix has a numerical value, for instance we use kilo to represent 1000, micro to represents 10-6 , i.e. 20 kilowatts = 20 x 1000 watts = 20 000 watts.
Sometimes prefixes may be represented using a factor or symbol as shown in table 2.
Table 2: Multiples and sub-multiples of SI units

Multiples and Sub-Multiples of SI Units
As you may see from table 2, multiples and submultiples are used to shorten long numbers e.g. we can 20 000 000 000 bites as 20 x 109 = 20gigabites =20 GB.
Conversions
After looking at multiples and sub-multiples, we can now convert SI units from one unit to another.
We use several conversion factors to translate a measurement from one unit to another unit. In table 4, we are translating units from column A to B by multiplying a unit in column B with a factor. For instance to change 1mile into a meter, you have to multiply a meter with a factor of 1609.344.
In cases where a unit cannot be translated directly to other unit, we translate it first to intermediate unit/s and then proceed to find our desired unit.
Table 3: Unit Convertion
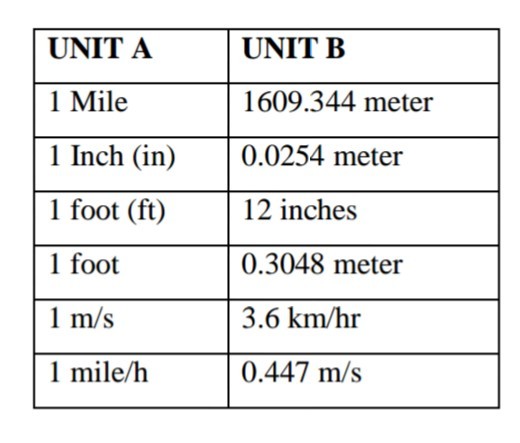
Units Convertion
Dimensions and Dimensional Analysis
A dimension is a physical variable used to specify the behavior or nature of a particular system. E.g. the length of a rod is so many meters.
We use square brackets [ ], to denote dimensions. For example we can represent the dimension of velocity as [V]=[L]/[T]
So far we have defined a dimension, but in most cases we use dimensions to express a dimension. The dimensions of a physical quantity are the powers to which the basic quantities (M, L and T) must be raised to represent a physical quantity. We represent a basic quantity, which does not appear in the physical quantity by raising it to power zero.
Example 2.
Find the dimension of acceleration a, and the dimensions of M, L and T in the dimensional formula of acceleration.
The dimension of acceleration; [a] = [M0LT–1]/[T]= [M0LT–2]
Hence the dimensions of acceleration are 0 in mass, 1 in length and -2 in time. Alternatively we can present dimensions of acceleration as M = 0, L = 1 and T = -2.
Dimensional Analysis
Dimensional Formula
This is the expression of a physical quantity in terms of its dimensions. For example, the dimensional formula for force is
[F]= [MLT–2]
Dimensional Equation
If you express an equation containing physical quantities, each quantity represented by its dimension formula, the resulting equation is referred to as Dimensional Equation.
Consider the formula v= u +at
Here u is initial velocity of the body, a is acceleration and t is the time taken to attain final velocity v. Writing this equation in the dimensional form, we have
[M0LT–1]=[M0LT–1] + [M0LT–1] + [M0L0T]
NOTE: in a dimensional equation, the dimensions in every term should be the same
TABLE 3. Dimensional formulae of Physical Quantities
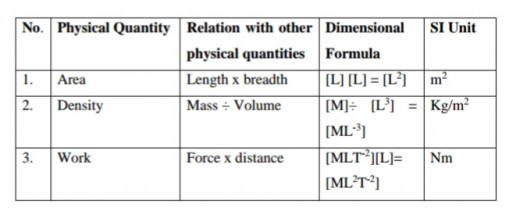
Dimension Formula of Physical Quantities
Categories of Physical Quantities
When you carefully study dimensional formula of the physical quantities you will discover that there are four categories of physical quantities.
a. Dimensional variables: Physical quantities that have dimensions but do not have fixed value e.g. force, velocity, work, power, etc.
b. Dimensional constants: Physical quantities which posses dimensions and have fixed value e.g. gravitational constant, Planck’s constant, etc.
c. Dimensionless variables: Physical quantities, which have neither dimensions nor fixed value e.g. Unfixed Constants: specific gravity, strain,
- Trigonometric functions: Cos 30°
- Logarithmic functions and exponential functions: log 10,
d. Dimensionless constants: Physical quantities, which do not posses dimensions but have a fixed value e.g.
- Numerical values: 1, 2, 200
- Fixed constants: π, index of refraction (n), etc.
Uses of Dimensional Formula (Analysis)
Dimensional analysis is used in the following ways:
- To check the correctness of an equation
- To remember a forgotten formula
- To derive relationship between different physical quantities
- To convert one system of units to another
- To find the dimensions of constants in a given relation
Checking the Correctness of a Physical Relation (Equation)
In order to check if a physical relation or a scientific equation is correct or not, dimensional analysis is applied. When the dimensions of the two sides of the equation thus, the right hand side (RHS) and the left hand side (LHS) are equal a scientific equation is considered as dimensionally correct and visa versa.
Example 3
Show if the equation, v = u+at2 is dimensionally correct or not. Where v is final speed, u is initial speed, a is acceleration and t is time.
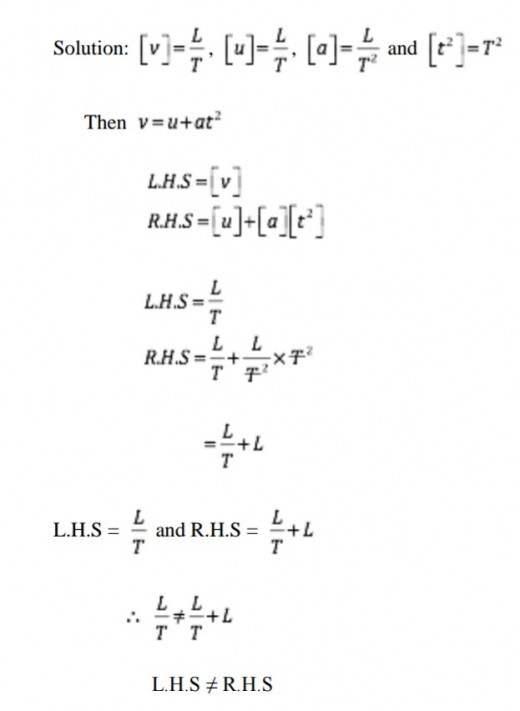
The equation is dimensionally incorrect.
To Calculate Dimensions of Unknown Quantity
When you want to find an unknown variable in an equation, you simply make it the subject of the formula and evaluate dimensions of the terms on the right hand side of the equation.
Example 4
A sphere of radius a, moves with velocity v, in a medium and the force F acting on it, is given as;

As you have observed from example 2, it is easier to work out SI units for an unknown quantity using dimensional analysis. Having found the dimensions of unknown quantity we can proceed to find SI units for unknown quantity. Each dimension gives a unit.
Measurements and Accuracy
Measurements
Measurement is the comparison of unknown physical quantity with a known fixed quantity.
For example if the length of the rod is 3 metres, it means the unit of length is the metre and this unit is contained 3 times in the length of that rod.
Two things represent the magnitude of a physical quantity;
i. The unit in which the quantity is measured e.g. metre
ii. The numerical value – this expresses how many times a unit is contained in a physical quantity, e.g. 3.
Accuracy
You may recall from your secondary school physical science that accuracy is defined as the measure of how close the measured value of a quantity corresponds to its true value. Oftenly we use the term accuracy hand in hand with precision. The degree of reproducibility or the closeness between repeated measurements is called precision.
Every measuring instrument has an inherent limit of precision or accuracy. This limit is determined by the list count of the instruments. No matter how carefully we measure, we can never obtain a result more precise than the limit of our measuring device. In experiments we calculate the limit of precision by considering ½ of the smallest division built into the device.
When you are using a vernier calliper that has a smallest unit of measurement as 0.1mm, its limiting precision is 0.1 x ½ = ± 0.05mm = ± 0.005 cm. Suppose using this vernier calliper, you have measured the length of a rod as 5.95cm, your limit of precision will be the same ± 0.05cm. The length of the lord will be represented as 5.95±0.05 cm. This means the true value of the length lies between 5.95 – 0.05 cm (5.9 cm) and 5.95+0.05 cm (6.0 cm).
Significant figures
The number of digits in a measurement about which we are certain, plus one additional digit which is uncertain are known as significant figures. The greater the number of significant figures obtained in a measurement, the more accurate is the measurement. But in most cases the number of significant figures is limited by a limit of precision.
Rules for significant figures
When we are using significant figures we consider the following rules:
i. All non-zero digits are significant e.g. 157.23 g contains five significant figures
ii. All zeros between two nonzero digits are significant e.g. 305.008 m contains six significant figures
iii. Unless stated otherwise, all zeros to the left of an understood decimal point but to the right of a nonzero digit are not significant e.g. 904,000s contains three significant figures.
iv. All zeros to the left of an expressed decimal point and to the right of a nonzero digit are significant e.g. 406.000 contains six significant figures.
v. All zeros to the right of a decimal point but to the left of a nonzero digit are not significant e.g. 0.000132ml contains three significant figures.
vi. All zeros to the right of a decimal point and to the right of a nonzero digit are significant e.g. 0.08040N and 50.00N each contain four significant figures.
Calculations involving significant figures
You should always remember that mathematical calculations cannot increase either the precision or the number of significant figures of the measured quantities. When we are doing calculations involving significant figures, we apply the following rules:
a. In addition and subtraction, the number of decimal places in the result should be equal to the smallest number of decimal places of any term in the sum or difference of two terms.
b. In multiplication or division, the number of significant figures in the result should be equal to the number of significant figures of the list least precise term in multiplication or division.
Example 5
Evaluate the following:
i. 3.76m+46.855m+0.2m
= 50.815m
Applying the first rule, the position of a decimal place in the result should be the same as in 0.2.
Answer: 50.8
ii. 11.3m x 6.8m
= 76.84m2
Applying the second rule, the number of significant figures in the result will be the same as in 6.8m
Answer: 77m2
Error
An error (inaccuracy) in measurements of any sort is a departure of the output of any measuring system from the value truly representing the quantity being measured.
This departure occurs in practice, no matter how sophisticated the measuring equipment is. We usually define error as the uncertainty in measurement.
Types of errors
1. Systematic errors: These are errors that appear in measurement due to known causes. They include all inaccuracies that tend to be more in one direction than in the opposite one. We can reduce systematic errors by repeating measurements a number of times. Some important causes of such errors are:
- Incorrect design or calibration of the instrument
- Limitation of the method used for the measurement
- Incorrect reading or interpretation of the instrument
- Lack of accuracy of the formula being used
2. Random errors: These are errors which appear in a measurement due to unknown causes. Random errors have no set pattern and their magnitude depends on the researcher’s knowledge of the apparatus used. Taking repeated readings of a quantity and averaging reduces random errors, but not systematic errors.
3. Gross errors: these are errors due to sheer carelessness of the researcher. They mostly involve human errors. Gross errors cannot be corrected but we may reduce them. In most cases we generate gross errors by:
- Recording the observations wrongly
- Reading the instrument incorrectly
- Entering wring values of observations in calculations
- Not caring for sources of error and precision.
Error Analysis
When dealing with errors in physics we oftenly use the following words; true value, absolute error, mean absolute error, relative error and percentage error.
A true value is the arithmetic mean of a large number of readings of a quantity. If during an experiment you have taken a1, a2, a3 ….. an as part of n different readings of a physical quantity in an experiment, then the true value of that quantity is
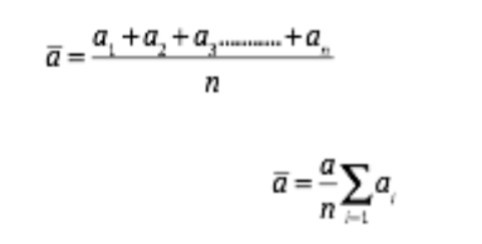
We note from the above equation that the true value is the same as arithmetic mean.
We define absolute error as the difference in the magnitudes of true value and the measured value of a physical quantity. We sometimes refer absolute error to actual error. If you observed a1, a2, a3 ….. an as you n different readings of a physical quantity in an experiment then the absolute errors are given as;

