Atomic Theory and the Structure of the Atom
Learning Outcomes
By the end of this article, you should be able to
- Define a theory
- Describe the structure of the atom.
- Calculate relative atomic mass of an atom
- Define elements and compounds
- Calculate relative molecular and formula masses of compounds
- Write electronic structures of elements
- Explain pure substances and mixtures
- Describe the periodic table of elements and their properties
What is a Theory
Scientific problems are always checked by experimental work. If a large number of experiments all give similar results, the latter are then summarized into a single statement known as a law.
Thus, a law is a summary of the results of a large number of separate experiments all giving the same conclusion. Further experiments are carried out to account for the law. This is done by proposing a hypothesis.
If the results of the experiments agree with the hypothesis, those results are stated into what is called a theory.
Atomic Theory
The most important chemical theory was given by John Dalton (1766-1844). Initially, Dalton stated the hypothesis that all substances consist of small particles of matter of several differentkinds corresponding to the different elements.
He called these particles atoms (from Greek: 'atomos' which means indivisible).
Dalton also said that elements consist of atoms and that atoms of one element are identical and compounds result from the combination of atoms of two or more elements each in a definite number.
He assigned a weight of 1 (one) to a hydrogen atom and built a table of relative weights of elements and compounds.
This hypothesis was verified by further experimental work and so it became a theory. Thus Dalton's atomic theory can be summarized into the following statements:
a) All matter is made up of atoms that cannot be created or destroyed; that is, atoms are indivisible by ordinary chemical means
b) Atoms of a particular element are identical in size, shape, mass and all other properties and differ from other atoms in these properties.
Experiments that were carried out later showed that atoms of one element may have different masses depending on the number of neutrons present.
Atoms of one element having different masses are called isotopes. You will learn more about isotopes. For now you should realize that statement (b) is not very correct.
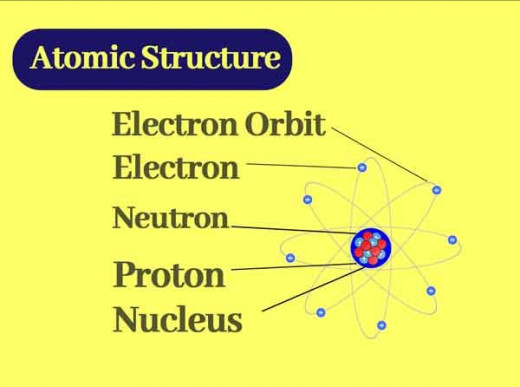
Structure of the Atom
In Science, people are always curious. Having accepted that an atom exists, the Scientists then wanted to know how this atom looks like. As such, they carried out several studies.
From these studies, it was found that the atom has even smaller particles which are called subatomic particles.
For example it was found that an atom consists of a positively charged nucleus surrounded by light (not heavy) negatively charged particles called which were named electrons.
In 1909, Ernest Rutherford studied the deflection patterns of alpha particles (He2+ ions) by thin metal foils and an analysis of the results led him to the conclusion that an atom has a tiny central nucleus that carries practically all the mass of the atom and that the lighter particles (the electrons) are around this nucleus. The number of positive charges on the nucleus is called the atomic number (abbrev. Z).
However, since the atom was found to be neutral, it was clear that the number of positively charged particles must equal the number of negatively charged particles (electrons).
Therefore, the number of electrons surrounding the nucleus in a neutral atom, must also be equal to the atomic number. The positively charged particles are called protons. The hydrogen atom for example, has one proton in its nucleus and carries one electron.
Furthermore, it was discovered that the electrons are arranged around the nucleus in ''shells''. Imagine this on a magnified scale.
You put the first group of electrons on a small ''circle'' with the nucleus inside it. Then put the second group of electrons on a bigger circle with the smaller one inside it and so on.
Figure 1.1 illustrates this pattern.
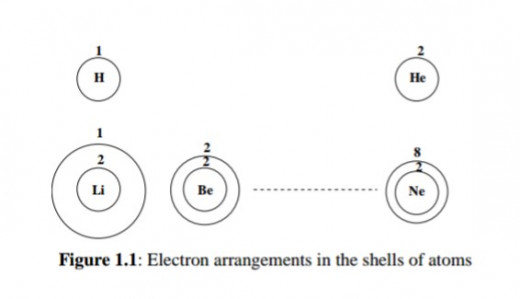
Electron Arrangements in the Shells of Atoms
Thus the successively added electrons first fill the innermost shell, then the next shell to it and so on. The innermost shell is called the K shell, the other shells in order of increasing size are called L, M, N, and so on.
We shall see later that the K shell is the lowest in energy, the L shell is higher in energy than the K shell, the M shell is higher in energy than the L shell and so on. We will advance the structure of the atom later.
Relative Atomic Mass (RAM)
We have stated that an atom has some components. The electrons are negatively charged and each has a charge of -1.60206 × 10-19 coulombs and a mass of 9.109 × 10-28g. The electron charge is abbreviated 'e' (-1.60206 ×10-19 = e) so that one electron has a charge of -e or just -1.
It should be noted here that the minus (-) sign does not mean that the electron is short of electric charge but that its electrical charge is of the type called negative. The protons are positively charged and each has a charge of +1.60206 × 10-19 coulombs and a mass of 1.672 × 10-24g.
As for the electron, the charge on a proton is simplified to +1. All the atoms except hydrogen also have neutrons which are neutral (have a charge of zero) but have a mass of 1.675 × 10-24g. The other components of an atom include neutrinos, positrons, μ-mesons and pi-mesons among others but the detailed nature of these is beyond the scope of this article.
The absolute masses of atoms cannot be determined as the atoms are very light. Only the relative masses can be obtained. To get these relative masses, we need to assign a weight to one atom (the standard) and relate the masses of the others to it. What we get then is the Relative Atomic Mass (RAM) of the atom.
Initially, hydrogen which had been assigned a mass of one, was chosen as the standard. However, for some practical reasons a carbon atom with six protons and six neutrons (carbon-12) was later chosen as the new standard.
The carbon-12 has a mass of exactly 12. The atomic masses of all the atoms were obtained with reference to a carbon-12 as the standard.
However, with technological revolution in modern times, mass spectrometers are used to determine the relative masses of atoms directly. Note that the relative atomic masses are rations hence have no units.
Since there are more than one atom for a particular element with differing relative masses we need to calculate a weighted mean of their masses taking into account their relative abundances.
This weighted mean for the mass of a particular element is called the atomic weight of that element. Therefore, we define the atomic weight of an element as the weighted mean of the relative masses of the isotopes of that particular element.
For example, chlorine has two isotopes with the relative masses and abundances of 35 (75.5%) and 37 (24.5%). Thus the atomic weight (the relative atomic mass) of chlorine is:
[ (35 × 75.5)/100] + [(37×24.5)/100]=35.4 9
Note in the example above that there is no chlorine in nature with a mass of 35.49. The atomic unit is called the Atomic mass unit (amu, given the SI unit, μ). One atomic mass unit is equivalent to1.66×10-24g (i.e., 1 amu = 1.66×10-24g ). Using this basis of measurement, the mass of an electron is 9.109×10-28g ÷1.66×10-24 g/amu = 0.000548 amu and that of a proton is 1.672×10-24g ÷ 1.66×10-24g/amu = 1.00725 amu and that of a neutron is 1.675×10-24 g ÷ 1.66×10-24g/amu = 1.00861 amu.
We usually round off atomic mass units to whole numbers called Mass Numbers (abbrev. A). This means that the mass of an electron is 0 while that a proton and a neutron each has a mass of 1.
The mass number (A) of an atom is the sum total of the number of protons and neutrons (since electrons are so light their contribution to the total mass is ignored). If we abbreviate the mass of neutrons N, then we have:
A =Z + N
where A, is the mass number, Z is the number (= mass)of protons
and N is the number (= mass) of neutrons.
It is possible to calculate the weight of an atom in grams or kilograms. Suppose, for example that 6×10-23 hydrogen atoms weigh 1g, what is the weight of one hydrogen atom?
Simple proportion shows that one hydrogen atom weighs 1g ÷ 6×1023 = 1.66×10-24g.
Note that the value obtained equals the weight of a single proton as given earlier. Since one hydrogen atom also weighs 1 amu, this confirms the fact that 1 amu= 1.66×10-24g as given above.
However, we don’t usually express atomic weights in grams or kilograms as the values become cumbersome in metric units.
Since 6 ×10-23 hydrogen atoms weigh 1g and one hydrogen atom weighs 1 amu, it follows that 1g = 6 ×1023amu. The figure 6×1023 (or more correctly 6.0226×1023) can be used to convert grams to atomic mass units. It is called the Avogadro's Number.
Isotopes
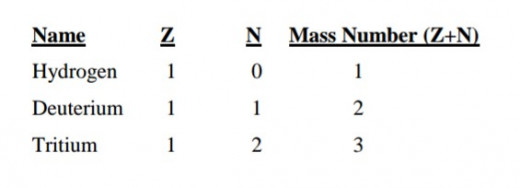
Atoms of a particular element all have the same atomic number and therefore, the same number of protons. Most of the elements do not have all their atoms with the same number of neutrons.
Such atoms belonging to the same element but having different numbers of neutrons in their nuclei are called isotopes.
It is clear therefore that isotopes have different masses. This is indeed the case as shown by the example below for the isotopes of hydrogen.
Elements and Compounds
Table 1.1 shows the names of some elements and their symbols.
Table 1.1: Some elements and their symbols

Some Elements and Their Symbols
Note that the first letter in the name of some few elements is taken as a symbol of that particular element. For example, hydrogen is H, carbon is C, nitrogen is N and so on.
However, because there are many more elements than the number of letters in the alphabet, some elements have two letters in their symbols. For these, the first letter is always in capitalized and the second one is written in small letter.
For example, chlorine is Cl, magnesium is Mg, neon is Ne and so on. In the foregoing examples, the first letter is derived from the English name. In other cases, the symbols of the elements are derived from Latin or German names.
For example, Antimony is Sb (from Latin, Stibnium), Copper is Cu (from Latin, Cuprum), Iron is Fe (from Latin, Ferrum), Sodium is Na (from German, Natrium), and potassium is K (from German, Kalium), sodium is Na (from German, Natrium) and potassium is K (from German, Kalium).
A compound is formed when atoms of two or more different elements combine. Examples of some compounds include water (H2O), methane (CH4), glucose (C6H12O6) and so on.
The kind and number of atoms in a compound are described by a formula as shown in brackets after the names of those compounds.
The usage of a formula can also be extended to an element if it naturally exists as a molecule. An example is dichloride (Cl2).
A formula indicates the relative numbers of atoms in a substance. There are three types of formulae; empirical formula is the simplest and gives a bare minimum information about a compound.
Diamond for example, consists of an array of carbon atoms each covalently bonded to four other carbon atoms but the formula of diamond, C, does not indicate this.
The other empirical formulae include, among others, NaCl (sodium chloride) and SiC (silicon carbide). In the general empirical formula of a compound, AxBy (where A and B are the atoms combined), the subscripts x and y are usually unity.
The second is a molecular formula, which has more information about a compound than an empirical formula. In the general formula AxDy, (where A and D are the atoms combined), the subscripts x and y in a molecular formula, are the actual numbers of atoms in the compound. For example, water has two hydrogen atoms and one oxygen atom; its molecular formula is therefore H2O.
The third type is a structural formula which shows how the atoms are joined to each other. In this case, the dashes are used to represent bonds and the atomic symbols represent atoms. For example, the structural formula of water is H-O-H.
Relative Molecular and Formula Mass
In the Section above, it was mentioned that compounds that exist as discrete entities, are described by a molecular formula. Such compounds are called molecules. Since the masses of atoms are relative, the masses of molecules are also relative.
Each molecule has its own relative mass which is the sum of the relative atomic masses of the atoms in that molecule. We therefore define the Relative Molecular Mass (RMM) as the sum of the relative masses of the atoms in a molecular formula.
For example:
(a) H2O has two hydrogen (H) atoms and one oxygen (O) atom
Relative molecular mass of water = (2× relative atomic mass of H) + (relative atomic mass of O)
= (2 × 1) + 16
= 18
(b) N2 has two Nitrogen atoms
Relative molecular mass of nitrogen molecule = (2× relative atomic mass of N) = 2 × 14
= 28
(c )C6H12O6 (glucose) has six carbon (C) atoms, 12 hydrogen (H) atoms and 6 oxygen (O) atoms
Relative molecular mass of glucose = (6 × relative atomic mass of C)
+(12 × relative atomic mass of H + (6 × relative atomic mass of O)
= 72 + 12 + 96
= 180
Compounds that do not exist as discrete entities however, do not form molecules and are represented by empirical formula.
For these we do not assign a Relative Molecular Mass; instead a Relative Formula Mass (RFM) is used. The relative formula mass is defined as the sum of the relative masses of the atoms in an empirical formula. For example:
NaCl has one sodium (Na) atom and one chlorine (Cl) atom
Relative formula mass of NaCl = (1× relative atomic mass of Na + (1× relative atomic mass of Cl)
= (1×23) + (1×35.5)
= 58.5
Pure Substances and Mixtures
We define substance that contains only one kind of compound or element as a pure substance. Take a spoon full of sugar, for example, this spoon contains only sugar molecules; therefore sugar is a pure substance.
Similarly, take copper metal; this consists of only copper atoms. Copper is also a pure substance. Pure substances have got the same, definite composition. This means that the concentration of the substance in a pure compound is constant.
When two or more different compounds (or elements) are put together and cannot react with each other, then we have a mixture.
Air for example, contains oxygen, nitrogen, water, carbon dioxide among others. It is a mixture of gases. Unlike pure substances, mixtures do not have definite compositions. The composition of substances in air, for example, may vary depending on weather conditions.
There are two types of mixtures; those in which the particles of each component are visibly present are called heterogeneous mixtures. An example is a mixture of say, salt and black pepper.
In this mixture, you can see the salt particles (white) and the pepper particles (black). The second type of mixture is where the particles of one component are evenly dispersed amongst those of the other.
This is called a homogeneous mixture. It is also called solution. Sugar or salt dissolved in water; methanol mixed with water; air on its own, are some examples of solutions.
Quantum Theory
We started on the subject of atomic structure in Section 1.3. We will now go a step further with this subject in this section. It is common experience than when you hit a football, it will move to any distance depending on the amount of energy you give it. Consider Figure 1.2a as an example.
The ball which is hit from position A, can stop at any of the dark points indicated. In this case, we say that the energy of the ball is continuous; That is, the ball can have any amount of energy.
If you hit the ball with less force it will gain less energy and so will move a short distance, if you hit it with more force it will gain more energy and likewise move a longer distance.
The motion of the ball here, is a part of the physics called classical physics.Newton'sLaws of motion are part of this physics. In reality, laws of classical physics apply to large particles such as the ball, the motion of the earth round the sun, automobiles, etc.
Figure 1.2: Comparison of a football having any amount of energy (a) (classical physics) and another one having only enough energy to take it from one point to the next (b)(quantum
physics)
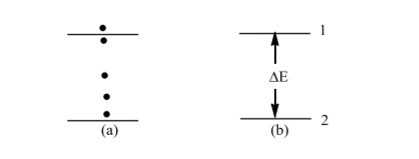
When classical physics was applied to atoms and other smaller particles, the results obtained were contrary to those expected. In 1900, Max Planck solved this mystery by stating that the energy of an object is not continuous; and that the object will only acquire enough energy to take it from one energy level to another.
Figure 1.2b illustrates this. If we give energy to an object in state 2 the object will only move to state 1 if it has acquired enough energy equal to ΔE = E1-E2(where, ΔE is change in energy, E2 is the energy of state 2 and E1 that of state 1).
Note in this case, the energy is in form of a packet or bundle and so, we speak of the energy being quantized. Thus the object can only have a certain definite energy value. Planck called this packet of energy (=ΔE) a quantum of energy.
If the energy of the object in energy level 2 is less than ΔE, the object will not move. This then started a new branch of physics called quantum physics whose laws apply to tiny particles like atoms, electrons, etc.
In 1013, NeilsBohr applied quantum theory to motion of electrons in atoms. He said that electrons move around the nucleus in circular orbits and as they do so, they emit or absorb energy that is quantized (it is now known that electrons do not actually behave this way).
The energy of an electron is governed by an integer called the Principal Quantum Number (n). The principal quantum numbers are just arbitrary numbers which are introduced in order to make predictions match experiment. They are given numbers 1,2,3,4, etc, increasing as an electron moves further away from the nucleus. In section above, we stated that electrons are in shells.
In simple terms, the principal quantum numbers are the shell numbers; The shells are numbered starting from the inter most as 1 and proceed as you move away. Thus the K shell is principal quantum number 1, L shell is principal quantum number 2 and so on.
Now the results of the experiments carried out later showed that under certain circumstances an electron behaves as a particle and under other circumstances it behaves as a wave just like a water wave. You will realise that one cannot locate the position of a wave since a wave extends in space.
We can only state that the chance of finding the wave at a particular point will be this much. In the same way, an electron being a wave, we can only state the chance of finding it at a certain place. The modern quantum theory says that electrons move around the nucleus in a definite region of space.
The region of space in which the electron spends at least 95% of its time is called an orbital. The orbitals are arranged in shells around the nucleus. For each principal quantum number (n), there are n2 orbitals.
These are shown in Table 1.2. Remember that numbering starts from the innermost shell since the K shell is shell number 1(that is principal quantum number 1).
Table 1.2: Shell symbols, orbitals and the maximum number of electrons in them for each principal quantum number
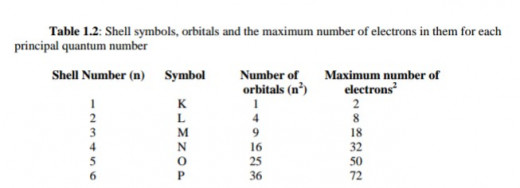
Shell Symbols, Orbitals and Maximum Number of Electrons
Apart from the K shell which has one orbital (the s orbital), both theory and experiments have shown that the group of orbitals in each of the L, M, N, etc., shells do not have the same energies especially in a many-electron atom.
In a hydrogen atom, which has only one electron, the orbitals in each group have the same energy. Such orbitals having the same energy are said to be degenerate. In a many-electron atom, the L shell has two energy levels; the lower one has one orbital which is called an s orbital, the higher one has three identical orbitals which are degenerate and these are called p orbitals.
The s orbital is spherical whereas the p orbitals are dumb-bell shaped situated at right angles to each other and in three dimensions along the x-, y- and z-axes; hence designated npx, npy and npz respectively, (where n is the principal quantum number).
The M shell has three energy levels; the lowest one consists of one orbital (also an s orbital), the second one has three orbitals which are identical and degenerate (also called p orbitals); and the third level consists of five degenerate orbitals called d orbitals. The N shell has four energy levels, s, p and d just like the M shell, but in addition, the highest one has seven degenerate orbitals called f orbitals.
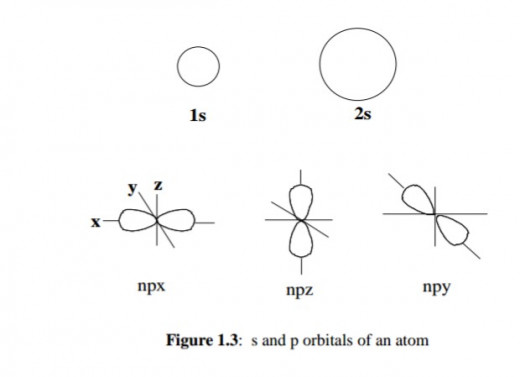
s and p Orbitals of an atom
Therefore, within a given shell, the energies increase in the order s<p<d<f3, etc. It was stated earlier that the energy of the electron is governed by the principal quantum number n; this number also governs the size of the orbitals.
As shown in Figure 1.3, an s orbital of principal quantum number one (denoted 1s) is smaller than an s orbital of principal quantum number two (denoted 2s).
As we shall see in the next section, the orbitals can be specified by the ''n'' values in which they appear; thus we would talk of a 1s orbital to mean an s orbital of principal quantum number one.
Similarly, a 2s or 3s orbital would mean an s orbital in principal quantum numbers two and three respectively. Analogously, 2p orbitals are those of principal quantum number two (2px, 2py or 2pz).
In the same way, we can also specify an electron in a particular orbital. Thus a 1s electron or a 3d electron means an electron in the s orbital of principal quantum number one or an electron in the d orbital of principal quantum number three, and so on.
Electronic Structures
Electrons are added to each atom, one at a time across a row. The lowest energy shell fills first then the next and so on. Within a shell, The lowest energy orbitals fill first and once those are full, the next lower energy ones fill and so on.
Structures which show the n-values and their orbitals that are occupied, together with the number of electrons in them are called electronic structures. The question we can ask at this point is: ''how many electrons can a single orbital accommodate?''.
The answer to this question was arrived at by Wolfgang Pauli, who introduced a restriction on the number of electrons which can go into a single orbital. The restriction called the Pauli Exclusion Principle states that ''any orbital will not hold more than two electrons''. This means that any orbital can hold 0, 1 or 2 electrons but not more than 2.
Therefore, the maximum number of electron allowed for each n value will be equal to 2n2 (see Table 1.2). When one electron occupies an orbital, it is said to be unpaired, when there are two electrons in an orbital, they are said to be paired.
Remember, in any atom the K shell is the lowest in energy followed by the L and then the M and so on. Within a shell, the s orbital is the lowest in energy followed by the p orbitals and then the d orbitals and so on.
The lowest energy shell and the lowest energy orbital in that shell fills first. With this and the Pauli Exclusion Principle in mind, we can now write the electronic structures for some of the atoms (Note: In order to write electronic structures of atoms you will need to refer to the periodic table of elements. One is included at the back of this module).
We begin with the simplest atom, hydrogen. This atom has got only one electron, and this will therefore, go into the s orbital of the K shell. The electronic structure we can write for hydrogen is therefore, 1s1.

The lowest energy level of an atom is called the ground state of that atom. For example, we have seen above that in hydrogen the electron normally resides in the 1s orbital.
The electronic state 1s1 is the lowest energy state for hydrogen and it is therefore the ground state. However, the electron in hydrogen can also move to higher levels, say to a 2s orbital. Such states of higher energy are called excited states of the atom.
Note that the s orbital of the K shell of hydrogen is half-full so that in the next atom, helium, an added electron will go into the same orbital and the electronic structure for helium is therefore 1s2.
At this point, the s orbital of the K shell is full in accord with Pauli Exclusion Principle. In the next atom, lithium (Li), the L shell starts filling and the electron will go into the s orbital of this shell (n = 2). The electronic structure of lithium is therefore, 1s22s1 and that of beryllium (Be) is 1s22s2.
The s orbital of the L shell is now filled up, and in the next atom boron (B), the p orbitals of the L shell start filling so that the electronic structure of boron is 1s22s22p1. Since there are three p orbitals, six electrons are required to fill them completely and the atoms B, C, C, O, F and Ne all fill up the 2p orbitals with Ne having the electronic structure 1s22s22p6.
The trend is the same in the third row of the periodic table. The s orbital fills first for this row (Note: this is the s orbital of the M shell) in Na and Mg, followed by the p orbitals. The d orbitals are not filled. In the fourth row, the s orbital of the N shell fills first (in K and Ca). In the next atom scandium (Sc), the d orbitals of row three become lower in energy than the p orbitals of row four, and so they start filling.
The electronic structure we can write for scandium is therefore, 1s22s22p63s23p64s23d1. The d orbitals require ten electrons (for the five of them) and the ten atoms Sc to Zn fill these orbitals. After the 3d orbitals are filled, the p orbitals of the fourth row start filling. Again, note that the d and f orbitals of the fourth row are not filled at this point.
The d orbitals for row four are filled in the fifth row after the s orbital of that row is filled. In the sixth row, the s orbital of that row fills first and then the next electron in La (atom 57) enters the 5d orbital after which, the f orbitals of row four (elements Ce-Lu) start filling.
These require fourteen electrons (for the seven of them) and so we see a group of fourteen atoms normally written at the bottom of the table. After the 4f orbitals are filled, the 5d orbitals continue filling in the remaining nine atoms (Hf-Hg; note that one 5d electron went into La) followed by the 6p orbitals.
The filling up of electrons into orbitals becomes complicated as we go down the table so that we will not continue any further. From the foregoing discussion, we note that the orbitals are filled in the order:
1s<2s<2p<3s<3p<4s3d<4p<5s<4d<5p<6s<4f<5d<6p....
The electronic structures are also written in the same order. There are three points you need to note in these structures. The first is that the sum of the superscripts in the electronic structure gives the atomic number of the atom.
For example, in the electronic structure of oxygen, 1s22s22p4, the sum of the superscripts is 8 (2+2+4), so the atomic number of oxygen is 8. This means that once you know the atomic number of an atom, you can easily write its electronic structure and vice versa.
The second is that the sum of the electrons in the highest principal quantum number, especially for the main group atoms, gives the group number of that atom. For example, in the electronic structure of oxygen, the highest principal quantum number is 2, and there are 6 electrons there (2 + 4 = 6). So oxygen is a group 6A element. Finally, the highest principal quantum number in the electronic structure gives the row (period) in which that atom appears.
In the example of oxygen, the highest principal quantum number in its electronic structure is 2, so oxygen is in row or period 2.
Hund's rule
The Pauli Exclusion principle stated earlier tells us exactly how many electrons can go into a particular orbital; an orbital will not have more than two electrons. With this principle, we can straight away write the electronic structure for example, carbon as, 1s22s22p2. In Section above, we saw that there are three degenerate p orbitals designated npx, npy, npz.
The question now arises: "how do the two 2p electrons of carbon enter the three p orbitals?". There are two possibilities; either both can go into the 2px orbital (i.e., 1s22s22px2, with the 2py and 2pz orbitals empty) or one can go into the 2px and the other one into the 2py (i.e., 1s22s22px12py1 with the 2pz orbital empty).
Friedrich Hund came up with a rule that is used to decide which electronic structure will be the most stable. The rule, called Hund's rule states that "electrons will occupy degenerate orbitals singly if those orbitals are empty".
This means that as long as there is a set of orbitals with the same energy, the electrons will go in separate orbitals until each such orbital has one electron. After that, additional electrons will start pairing up in accord with the Pauli Principle. With this, we can write the most stable electronic structure of carbon as: 1s22s22px12py1 (note: the choice of which orbital is occupied i.e., whether the 2px, 2py or 2pz, is purely arbitrary).
The electronic structures of nitrogen, oxygen, fluorine and neon are therefore: N: 1s22s22px12py12pz1, O: 1s22s22px22py12pz1, F: 1s22s22px22py22pz1, Ne:1s22s22px22py22pz2. Note that nitrogen has one electron in each of the p orbitals in accord with Hund's rule and that pairing starts in oxygen. The same trend applies to the d orbitals and also, the f orbitals.
Paramagnetism and diamagnetism
The electronic structure of carbon given above has two unpaired electrons in the p orbitals while nitrogen has three such electrons. Oxygen and fluorine have two and one unpaired electrons respectively, while neon has no unpaired electrons.
Elements such as carbon, with unpaired electrons, tend to be attracted by a magnet and are said to be paramagnetic. Elements such as neon with all the electrons paired tend to be repelled by a magnet and are said to be diamagnetic.