Particulate Nature of Matter
The Particulate Nature of Matter
The kinetic theory is a scientific model which helps us to explain how the arrangement of particles relates to the properties of the three states of matter, which are solid, liquid and gas. Chemistry helps us to understand and know how matter is like and how it behaves and this helps us to predict how different matters could behave under different conditions. Much as this is the case, the big question remains as what is matter? Matter can be defined as all substances and materials from which the physical universe is composed of, and they can be categorized as solids, liquids or gases
Kinetic Theory of Matter
As of today, there are numerous evidences which explains that all matter is made up of tiny particles and the kinetic theory helps us to explain the way in which different types of Matter behaves and this relate to physical properties of matter.
Key points of the Kinetic Theory of Matter can be summarized as follows:
- All the three states of matter are made up of tiny particles which move at a constant rate, and are invisible to the naked eye. It should be noted that different substances are made up of different types of particles, which are atoms, molecules or ions, which have different sizes.
- The particles in all the three states of matter move all the time and the rate at which they move depends on the amount of temperature such that the higher the temperature, the faster they move on average.
- The rate at which the particles move in all the three states of matter depends on weight of particles, thus the heavier particles the slower they and the lighter the particle, the faster they move.
Explaining the States of Matter
Solid
- The particles attract one another in solid and there are strong forces of attraction which hold them close together, and this makes them incompressible.
- Particles in solid have little freedom of movement unlike in the other state of matters and can only vibrate on a fixed position.
- They have a regular shape because they are arranged in a regular form and this explains why many solids form crystals.
- All solid, have a definite shape and volume at a given temperature, which may be affected by changes in temperature. Solids expand when heated and contracted when cooled.
Liquid
- Particles in liquid are close together, but they randomly move around and often collide with each other.
- Liquid has a weaker force of attraction comparing them to that of solid and particle in a liquid are known to have more energy than that of the solid.
- Just as in solids, particles in liquid expand when heated and contracted when cooled.
- In liquid at any given temperature, it tends to have a fixed volume and takes the shape of the container in which they are put. Any change in temperature, affects their volume, just like in solid.
Gas
- Unlike in solid and liquid, in a gas the particles are relatively far apart and they move freely anywhere within the container in which they are held and this makes them to fill the space in which they are put.
- Their particles move in a random order and they do so at very high velocities and more rapidly than those in a liquid.
- Particles in gas collide with each other, but not as often as in liquids.
- Their particles have no forces of attraction and are far apart, making them to exist virtually.
- At a given temperature, gas has neither a fixed nor a definite volume and it will correspond by taking the shape of the any container into which it is put and will spread out evenly within that container.
- Gases are much more compressible than liquids.
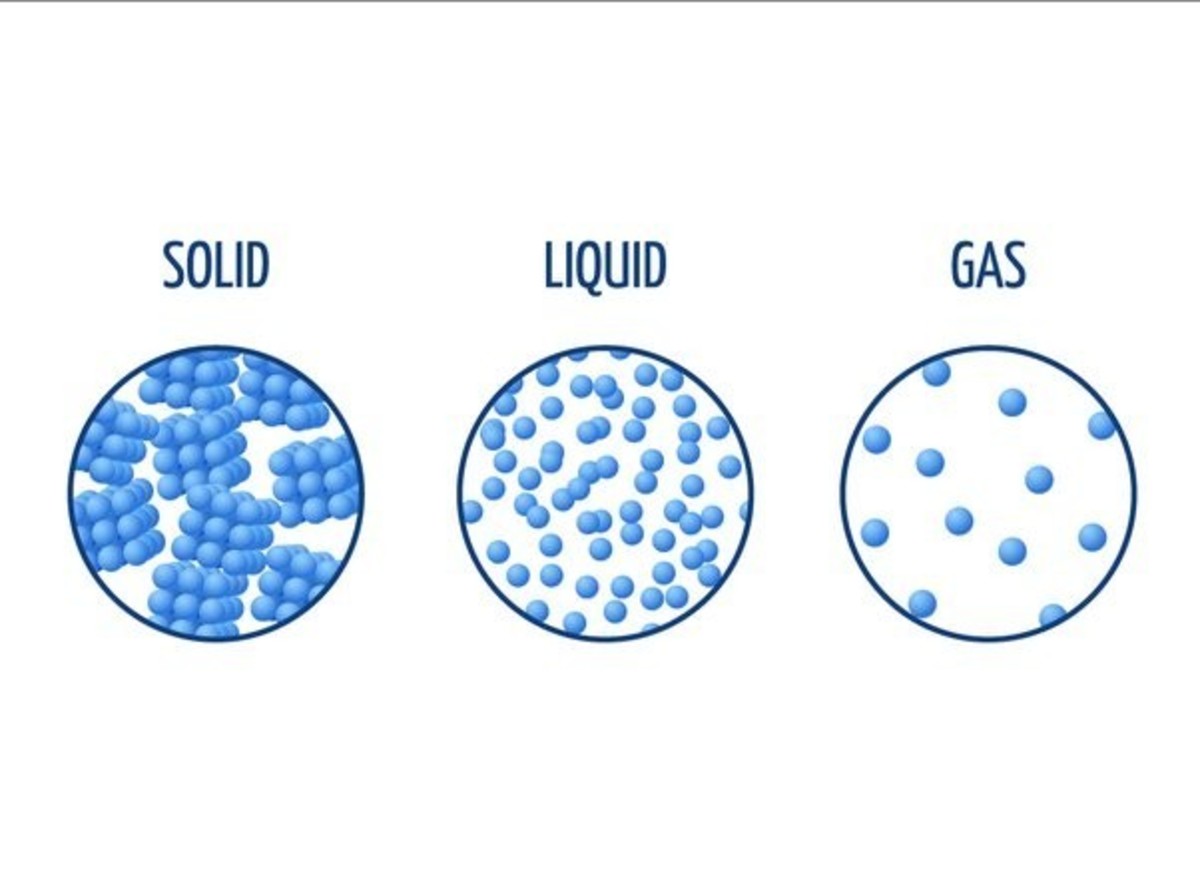
The arrangement of particles in solids, liquids and gases is shown in Figure 1.
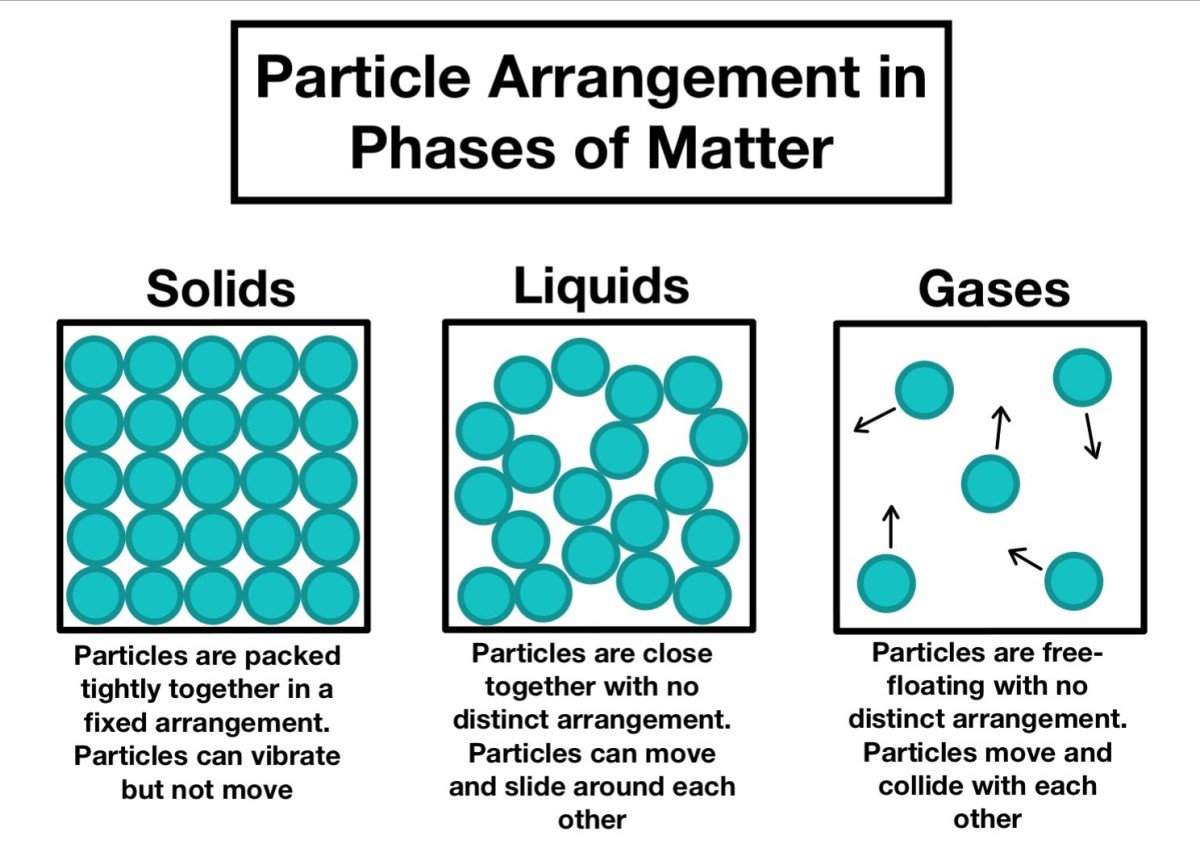
Arrangement of Particles in the 3 States of Matter
Changes of State
The kinetic theory is a model which helps us in explaining how a substance changes from one state of matter to another. For example, when a solid is heated it gains energy which makes the particles in it to vibrate faster and this makes their particles to be pushing each other. When their particles are pushing each other after gaining the heat energy, it causes an increase in their volume of the solid, making it to expand.
The heat energy which solid particles gain, weaken their forces of attraction and this makes the regular pattern structure in solid to break down and their particles can ably move around each other and at this state, the solid has melted and the temperature at which this takes place is called the melting point of the substance.
It should be noted that temperature of any pure melting solid will be constant until it has all melted. It should also be noted that when the substance has become a liquid, there are still very significant forces of attraction between the particles, which is why it is a liquid and not a gas. Some solids have a higher melting point and these solids with higher melting points tends to have stronger forces of attraction between their particles than those solids which have low melting points.
When the liquid is heated, their particles gain energy which makes them to move around faster as their average energy increases. As more heat energy is applied, particles at the surface of the liquid tends to gain enough energy which overcomes the forces of attraction between themselves and the other particles in the liquid, and this makes their particles to escape forming gas. A stage at which liquid forms a gas is called evaporation.
The temperature at which liquid particles escape to form gas is called boiling point. At the Boiling point the pressure of the gas created above the liquid equals that in the air – atmospheric pressure.
A liquid which has strong forces of attraction tends to have higher boiling point between their particles than liquids with low boiling points. In gases the average energy of the particles decreases when the gases get cooled and their particles move closer together.
Their forces of attraction between the particles now become significant and cause the gas to condense into a liquid. When a liquid is cooled it freezes to form a solid. In each of these changes energy is given out.
All the Changes of state of matter we have talked above are examples of physical changes. When a physical change of state occurs, the temperature remains constant during the change. It should be noted that during a physical change no new substance is formed in any state of matter.
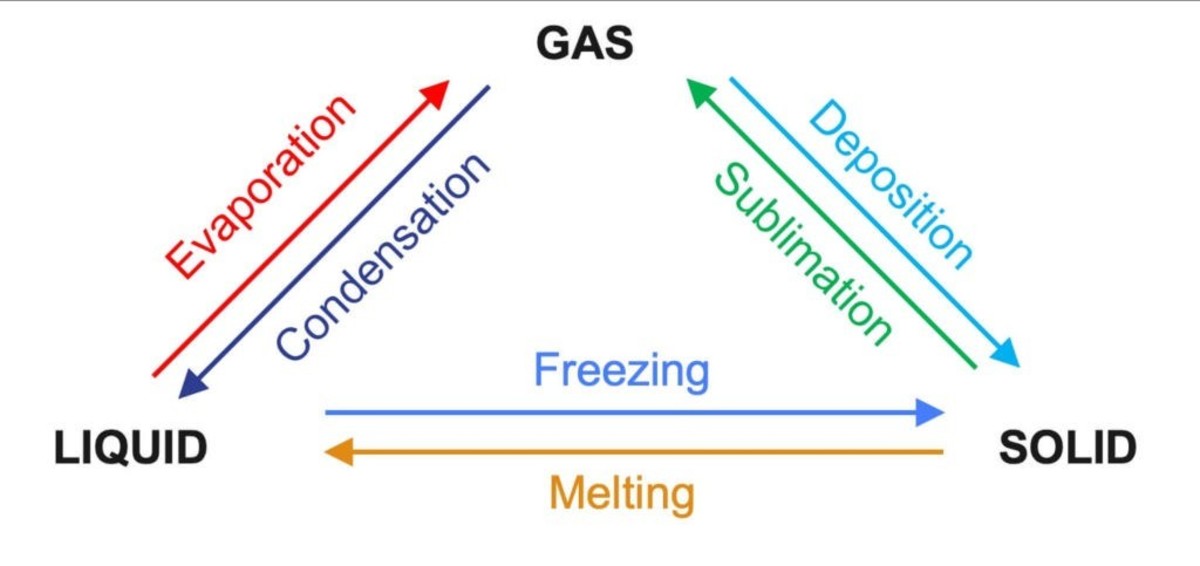
Changes in the States of Matter
Heating and Cooling curves
The graph below is the heating curve and was drawn by plotting the temperature of water against heat, as water was heated steadily from −15 °C to 110 °C. You could see from the curve that changes of state have taken place.
In the below graph when the temperature was first measured only ice was present. After a short time the curve begins to flatten, showing that even though heat energy is being put in it, the temperature remains constant.
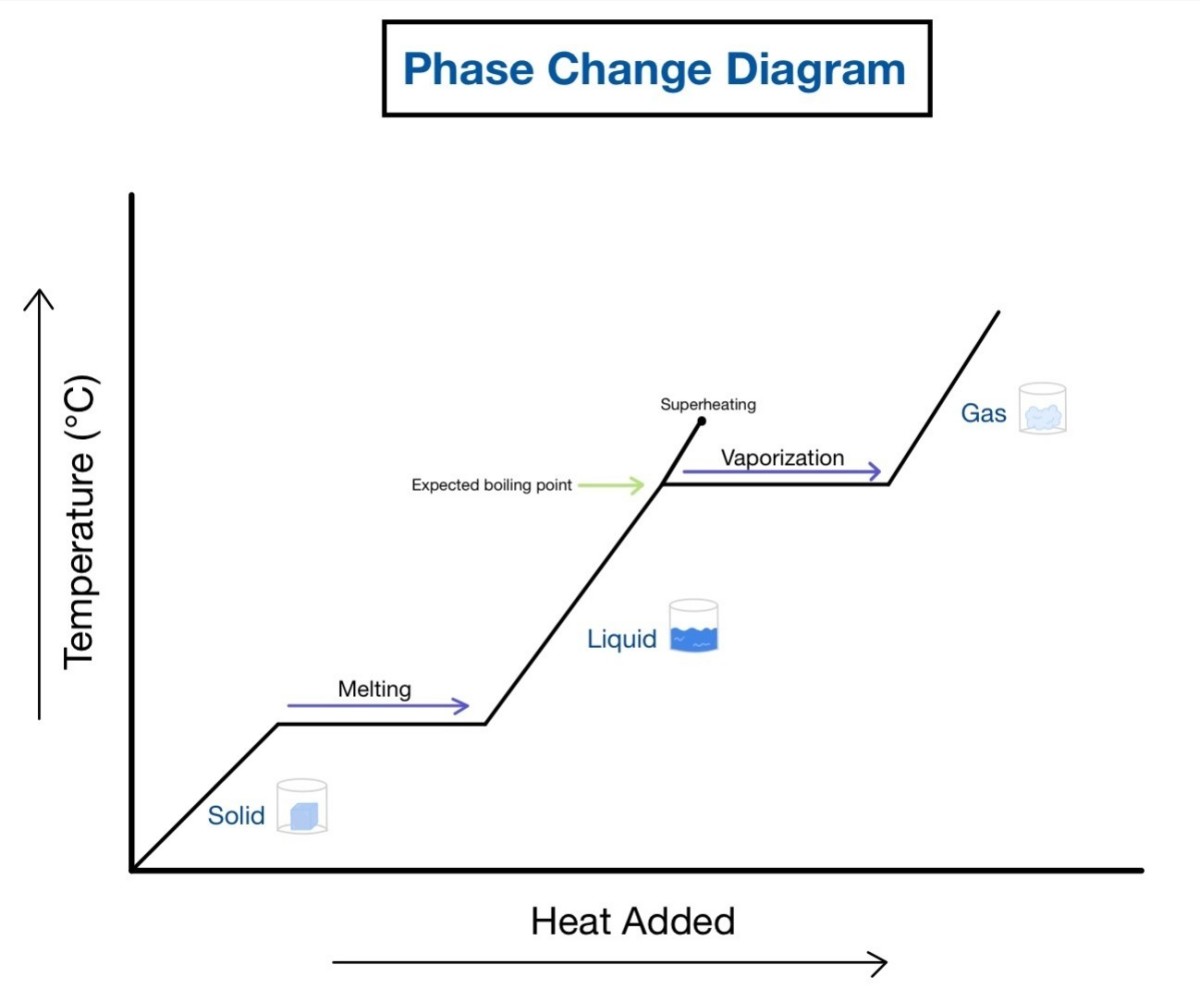
Heating Curve
Particles in ice of water are known to be close to each other and are attracted to one another. For the ice to melt, their particles need to obtain sufficient energy to overcome the forces of attraction between the water particles to allow relative movement to take place. This is where the heat energy is going in the above graph.
When all the ice have melted, the temperature will begin to rise again. Generally, the heating curve for a pure solid always stops rising at its melting point and gives rise to a sharp melting point. A melting point indicates a pure sample. The addition or presence of impurities lowers the melting point.
Useful Definitions
Absolute Zero
This is the theoretical temperature (which can never be reached) at which all particle motion stops. Absolute zero is -273°C or 0 Kelvin. For us to calculate it, we use this simple equation, which is, Kelvin = °C + 273. A 1K change equals a 1°C change.
Brownian Motion
This is the random motion of particles which is caused by smaller and faster moving water particles which are constantly colliding with them and moving them around. It should be noted that an observation of Brownian motion proves the correctness of the kinetic particle theory.
Diffusion
This is the process in which particles mix or spread by colliding randomly with each other, and bouncing off in all directions. Particles travel in random zigzag motions, this is how smells spread, solid dissolves, dust particles travel in a random way when suspended in air. All gases diffuse to fill the space available.

Diffusion
Evidence for diffusion
One of the evidences of diffusion is seen in a liquid potassium manganate (VII) which has been put in a beaker of water above. In the beaker above the colour of potassium manganate will spread as a result of Brownian/random motion.
Similarly, in gases: a gas jar of air and a gas jar of bromine in the figure above are connected, the bromine travels up the tube.
Factors that Affect the Rate of Diffusion
The following are some of the factors which affect the rate of diffusion.
Temperature increase
In particles the rate of diffusion increases with an increase in temperature. Similarly, when the temperature falls, the rate of temperature decreases.
2. Particle mass decrease
In particles the rate of diffusion increases when the mass decreases and vice versa (this is shown by the following experiment):
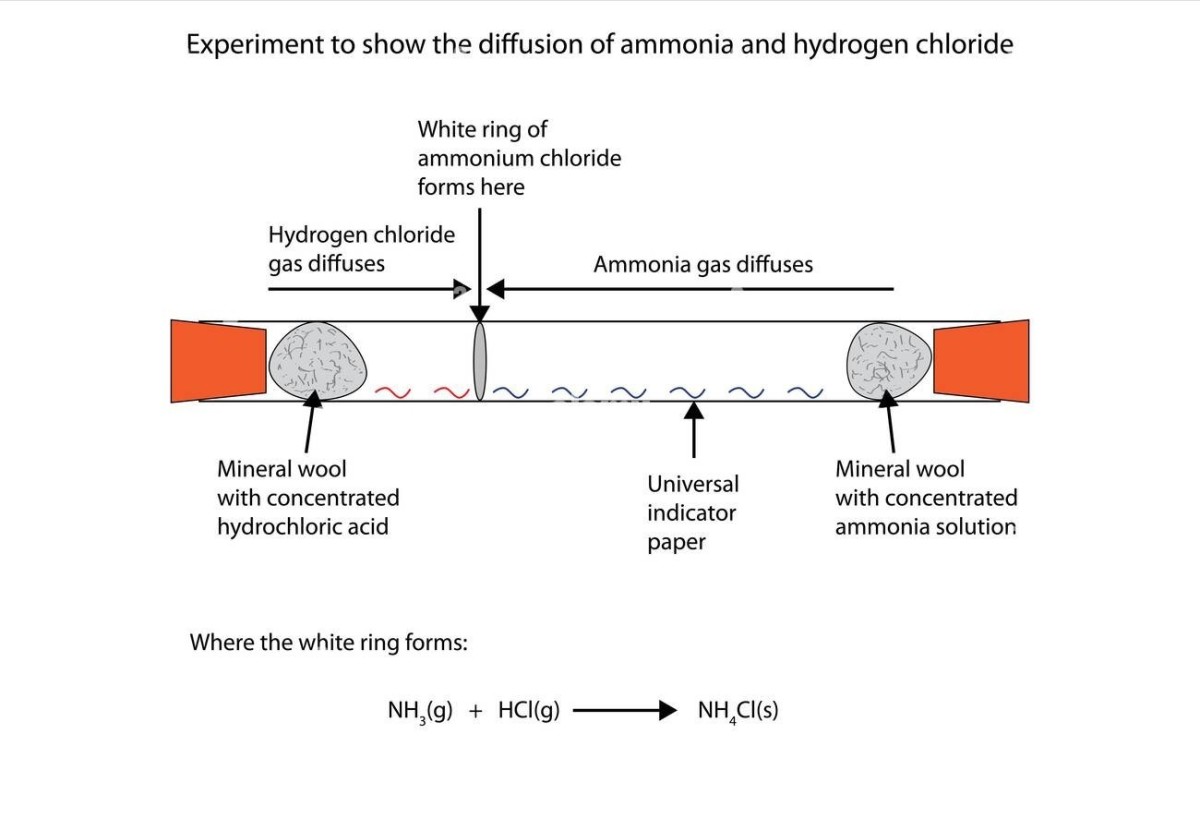
Evidence of Diffusion
From the figure above the NH3 molecules have less mass than the HCl molecule, so they diffuse faster, hence the product (a white cloud of NH4Cl) forms closer to the end where the HCl is.